UNIT 18: VARIATION IN TRENDS OF THE PHYSICAL PROPERTIES
Key Unit competence: Use atomic structure and electronic
configuration to explain the trends in the
physical properties of the elements.Introductory Activity 18
An extended family is a family that extends beyond the nuclear family,
consisting of parents like father, mother, their children, aunts, uncles,
grandparents and cousins. They all live in the same household. It is easily
to see characteristics that qualify as physical traits include hair color and
type, attached earlobes, adorable freckles, eye color and hairline among
family members.Think about this activity in order to understand well and answer the
following questions.1. Have you ever noticed physical similarities between relatives at a
family reunion?2. What do physical characteristics indicate among the family
members?3. What are the physical characteristics that may be possessed by the
same family members?18.1. Historical background of the Periodic Table
Activity 18.1
Use textbooks and internet connection and point out the main steps in
the development of the periodic table.Scientists have always been searching for patterns and similarities in the
properties and reactions of substances.For a long time chemists look at ways of dividing up the two big group,
metallic and non-metallic elements into smaller sub-groups. They grouped
together very reactive metals like lithium, sodium and potassium, slightlyless reactive metals such as calcium, strontium and barium and very reactive
non-metals like chlorine, bromine and iodine.Some elements were discovered which had properties in between metallic
and non-metallic. These elements were described as metalloids (e.g. silicon).• In 1669, Hennig Brand a German merchant and amateur alchemist
invented the Philosopher’s Stone; an object that supposedly could turn
metals into pure gold. He heated residues from boiled urine, and a liquid
dropped out and burst into flames. He also discovered phosphorus.• In 1680 Robert Boyle also discovered phosphorus without knowing
about Henning Brand’ discovery.• In 1809, curiously 47 elements were discovered and named, and
scientists began to design their atomic structures based on their
characteristics.• In 1829, Johann Dobereiner classified some elements into groups of
three, which he called triads.
The elements in a triad had similar chemical properties and orderly
physical properties.• Examples of triads : Cl, Br, I and Ca, Sr, Ba
• Dobereiner showed that when the three elements in each triad were
written in order of atomic mass, the middle element had properties in
between those of the other two.• In 1863, John Newlands (1838 - 1898) suggested that elements be
arranged in “octaves” because he noticed (after arranging the elements
in order of increasing atomic mass) that certain properties repeated
every 8th element. (Law of Octaves).His law of octaves failed beyond
the element calcium.• In 1869, Dimitri Mendeleev based on John Newlands’ ideas started
the development of elements organized into the periodic table. The
arrangement of chemical elements were done by using atomic mass
as the key characteristic to decide where each element belonged in his
table. The elements were arranged in rows and columns. He predicted
the discovery of other elements, and left spaces open in his periodic
table for them.• At the same time, Lothar Meyer published his own periodic table with
elements organized by increasing atomic mass.• In 1886, French physicist Antoine Becquerel first discovered
radioactivity. During the same period of 1886, Ernest Rutherford named
three types of radiation; alpha, beta and gamma rays.• In 1886, Marie and Pierre Curie started working on the radioactivity
and they discovered radium and polonium. They discovered that beta
particles were negatively charged.• In 1895, Lord Rayleigh discovered a new gaseous element named
argon which proved to be chemically inert. This element did not fit any
of the known periodic groups.• In 1898, William Ramsay suggested that argon be placed into the
periodic table between chlorine and potassium in a family with helium,
despite the fact that argon’s atomic weight was greater than that of
potassium. This group was termed the “zero” group due to the zero
valency of the elements. Ramsey accurately predicted the future
discovery and properties neon.• In 1913, Henry Moseley worked on X-rays and determined the actual
nuclear charge (atomic number) of the elements. He has rearranged
the elements in order of increasing atomic number.• In 1897 English physicist J. J. Thomson discovered small negatively
charged particles in an atom and named them as electrons; John
Sealy Townsend and Robert A. Millikan investigated the electrons and
determined their exact charge and mass.• In 1900, Antoine Becquerel discovered that electrons and beta particles
as identified by the Curies are the same thing.• In 1903, Ernest Rutherford proclaimed that radioactivity is initiated by
the atoms which are broken down.• In 1911, Ernest Rutherford and Hans Geiger discovered that electrons
are moving around the nucleus of an atom.• In 1913, Niels Bohr suggested that electrons move around a nucleus
in discreete energy levels called orbits. He observed also that light is
emitted or absorbed when electrons transit from one orbit to another.• In 1914, Rutherford identified protons in the atomic nucleus. He also
transformed a nitrogen atom into an oxygen atom for the first time.
English physicist Henry Moseley provided atomic numbers, based on
the number of electrons in an atom, rather than based on atomic mass.• In 1932 James Chadwick discovered neutrons, and isotopes werehe discovered plutonium and all the transuranic elements from 94 to
identified. This was the complete basis for the periodic table. In that
same year Englishman Cockroft and the Irishman Walton first split an
atom by bombarding lithium in a particle accelerator, changing it to two
helium nuclei.The last major changes to the periodic table give rise
from Glenn Seaborg’s work in the middle of the 20th Century. In 1940,
102.• In 1944, Glenn T. Seaborg discovered 10 new elements and movedout 14 elements of the main body of the periodic table to their current
location below the lanthanide series. These elements were known as
Actinides series.• In 1951, Seaborg was awarded the Nobel Prize in chemistry for hiswork. Element 106 has been named seaborgium (Sg) in his honor.• Presently, 118 elements are in the modern Periodic Table.Although Dimitri Mendeleev is often considered as the “father” of the periodic
table, however the work of many scientists contributed to its present form.The representation of a modern Periodic Table of Elements is shown below. The elements also belong to the families (chemical families). These elements
The elements also belong to the families (chemical families). These elements
have similar physical and chemical properties exist among the families and
the periods of the periodic table.Application activity 18.1
The periodic table is an important tool used in chemistry:
1. Why scientists have developed a classification of chemical
elements?2. Explain why the elements are classified in groups and periods of
the periodic table3. Choose one element of Group 1 and one of group 17 and make
their electronic configurations using orbitals.18.2. Comparison of Mendeleev’s table with the modern
Periodic TableActivity 18.21. Look at the modern periodic table and write down four things it tells
you.2. Explain the gaps found in the Mendeleev periodic table compared
to the modern one?3. How many elements does the modern periodic table contain?
The periodic table is the arrangement of chemical elements according to their
chemical and physical properties. The modern periodic table was created
after a series of different versions of the periodic table. The Russian Chemist/
Professor Dmitri Mendeleev was the first to come up with a structure for
the periodic table with columns and rows. This feature is the main building
block for the modern periodic table as well. The columns in the periodic
table are called groups or families, and they group together elements with
similar properties. The rows in the periodic table are called periods, and they
represent sets of elements that get repeated due the possession of similar
properties. The main difference between Mendeleev and Modern Periodic
Table are shown in the Table 18.1.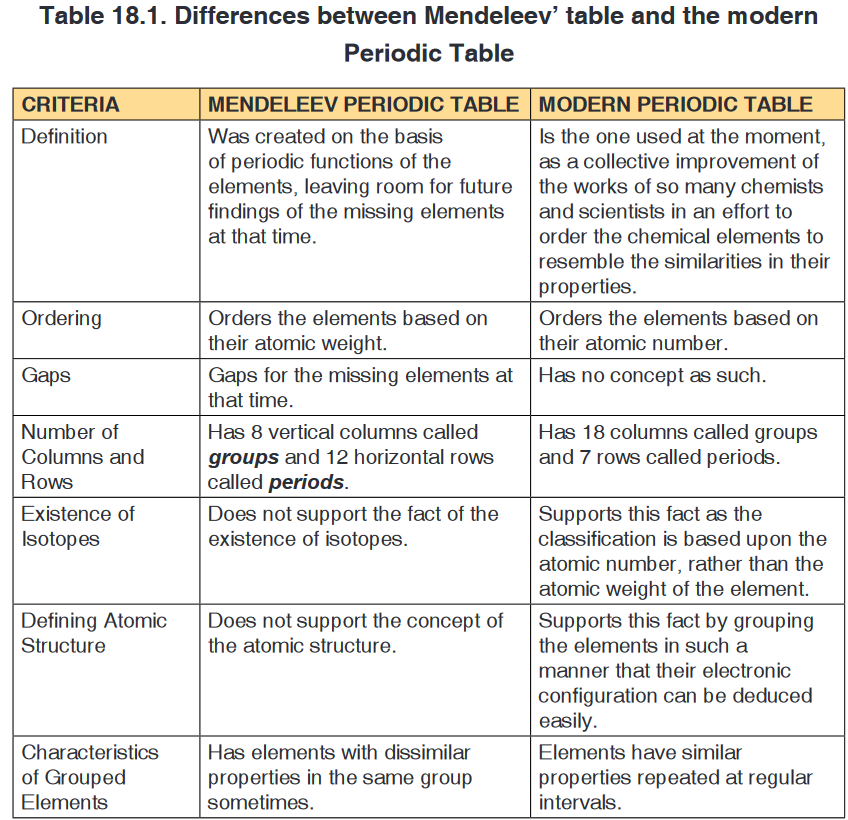 Application activity 18.2
Application activity 18.2
1. Discuss the similarities and differences of Mendeleev’s table and
modern periodic Table.2. How were the positions of cobalt and nickel resolved in the modern
periodic table?18.3. Location of the elements in the Periodic Table based
on the electronic configurationActivity 18.3
1. Based on knowledge gained in the previous years:
a) Represent the electronic configuration of the elements 25X and
11Y.
b) Discuss the information given by the number of electrons in
the last orbitals of the above element about their position in the
periodic table?
c) Explain the period and the group of the periodic table in which
the above elements are located.2. Is it possible to have an element with atomic number 1.5 between
hydrogen and helium?18.3.1. Major Divisions of the Periodic TableThe periodic table is a tabular of the chemical elements organized on the basis
of their atomic numbers, electron configurations, and chemical properties.In the periodic table, the elements are organized by periods and groups.
The period relates to the principal energy level which is being filled by
electrons. Elements with the same number of valence electrons are put in
the same group, such as the halogens and the noble gases. The chemical
properties of an atom relate directly to the number of valence electrons, and
the periodic table is a road map among those properties such that chemical
properties can be deduced by the position of an element on the table. The
electrons in the outermost or valence shell are especially important because
they participate in forming chemical bonds.18.3.2. Location of elements in modern Periodic Table usingexamplesIn the periodic table, the elements are located based on groups and periods.a. Finding Period of ElementsPeriod of the element is equal to highest energy level of electrons or principal
quantum number. Look at following examples for better understanding;

 Which one(s) of the following statements are correct, which one(s)
Which one(s) of the following statements are correct, which one(s)
are false for these elements.
a) X is alkaline metal
b) Z is halogens
c) U is lanthanide
d) T is noble gas
2. Explain the major parts of the periodic table?18.4. Classification of the elements into blocks (s, p, d,
f-block)Activity 18.4
1. Using the s p d f notation write condensed electronic configuration for
the following elements. Al (Z = 13), K (Z = 19), Sr (Z = 38) and Fe (Z
= 26). Br(Z = 35), Zr(Z = 40), Nd(Z = 60) ,Yb (Z= 70)2. For each element, identify the last subshell that is occupied by
electrons and draw adequate conclusions.The long form of periodic table can be divided into four main blocks. These
are: s- block, p- block, d- block, and f-block.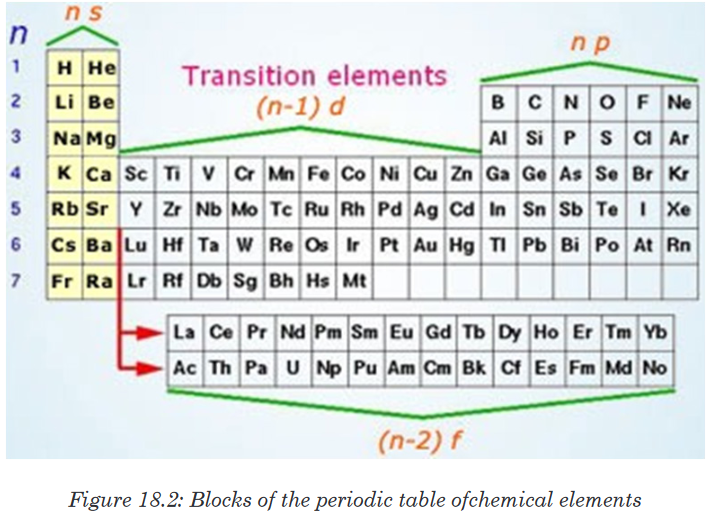 1. s-Block Elements
1. s-Block Elements
The elements in which the last electron enters the s-subshell of their
outermost energy level are called s-block elements.This block is situated at extreme left of the periodic table. It contains elements
of groups 1 and 2.Their general configuration is ns1-2, where n represents the outermost shell.The elements of group 1 are called alkali metals whereas the elements of
group 2 are called alkaline earth metals.2. p-Block Elements
The elements in which the last electron enters the p-sub-shell of their
outermost energy level are called p-block elements.The general configuration of their outermost shell is ns2 np1-4. The only
exception is helium (ls2). Strictly, helium belongs to the s-block but its
positioning in the p-block along with other group 18 elements is justified
because it has completely filled valence shell (1s2) and as a result, exhibits
properties characteristic of other noble gases. This block is situated at the
extreme right of the periodic table and contains elements of groups 13, 14,
15, 16, 17 and 18 of the periodic table.Most of these elements are non-metals, some are metalloids and a few
others are heavy elements which exhibit metallic character.The non-metallic character increases as we move from left to right across a
period and metallic character increases as we go down the group.3. d-Block Elements
The elements in which the last electron enters the d-subshell of the
penultimate energy level are called d-block elements.Their general valence shell configuration is (n-1) d1-10 ns1-2, where n represents
the outermost energy level. d-block contains three complete rows of ten
elements in each. The fourth row is incomplete.The three rows are called first, second and third transition series. They
involve the filling of 3d, 4d and 5d orbitals respectively. The d-block contains
elements of groups 3 to 12 of the periodic table.4. f-Block Elements
The elements in which the last electron enters the f-sublevel of the anti-
penultimate (third to the outermost shell) shell are called f-block elements.Their general configuration is (n–2)f1-14 (n–1) d0-1 ns2, where n represents the
outermost shell.They consist of two series of elements placed at the bottom of the periodic
table.The elements of first series follow lanthanum (57La) and are called
lanthanides. The elements of second series follow actinium (89Ac) and are
called actinides. Actinide elements are radioactive.Many of them have been made only in nanogram quantities or even less by
nuclear reactions. Chemistry of the actinides is complicated and is not fully
studied.Briefly, in the periodic table, elements are divided into:• The s-block (contains reactive metals of Group 1A (1) and 2A (2)),
• The p-block (contains metals and non metals of Group 3A (13)
through 8A (18)),
• The d-block (contains transition metals Group 3B (3) through Group
2B (12)), and
• The f-block (contains lanthanide and actinide series or inner
transition metals).Application activity 18.4
1. a) How many blocks into which chemical elements are classified and
how are they named?b) What is the criterion used for this classification?2. Which block of elements will tend to form: positive ions? Negative
ions?3. Give any two examples of elements of s block, p block, d block and
f block.4. Why d-block elements are called transition elements?5. Why f-block elements are called inner transition elements?18.5. Factors that influence the change of each physical
property of the elements across a period and down a
groupActivity 18.5The elements in the periodic table display many trends which can be
used to predict their physical properties.Use available resources and explain three of the factors that you think
can influence the physical properties of elements in the periodic table.In the Periodic Table, there are a number of physical properties that are not
really “similar” as it was previously defined, but are more trend-like. This
means is that as you move down a group or across a period, you will see a
trend-like variation in the properties.The actual trends that are observed with the physical properties have to do
with the following factors:
1. The number of protons in the nucleus (called the nuclear charge).
2. The number of electrons held between the nucleus and its outermost
electrons (called the shielding effect).Explanation:
Electrons in inner levels or shells tend to shield outer electrons from the full
nuclear charge, which is reduced to effective nuclear charge (Zeff).Electrons are held in an atom or ion by the electrostatic attraction between
the positively charged nucleus and the negatively charged electrons. In multi-
electron species, the electrons do not experience the full positive charge of
the nucleus due to shielding by electrons which lie between the electron of
interest and the nucleus. The amount of positive charge that actually acts on
an electron is called the effective nuclear charge.Electrons that have a greater penetration shield others more effectively. For
example, electrons in level n = 1 shield those in level n = 2 very effectively,
and those in n = 1 and n = 2 shield electrons in level n = 3. Electrons at the
same level, but in different sublevels, also shield other electrons to some
extent. The extent of penetration and shielding effect is in the order:s>p>d>f.The effective nuclear charge (Zeff) greatly influence atomic properties. In
general,• Zeff increases significantly across a period (left-to-right)
• Zeffincreases slightly down a group.Application activity 18.5Periodic Table Trends are influenced by the following factors:
a) Size of the atom
b) Nuclear charge
c) Shielding effect
Which of them affect the variation of physical properties in a group, in a
period?18.6. Variation of the physical properties down the group
and across the periodActivity 18.6
Analyze the table about the molar ionization energy values for some
elements and answers the questions that follow.
1. How is the variation of these values if you consider elements in the
same
a) period?b) group?
Using available resources including internet, attempt to give a
plausible explanation to the trend observed.2. Analyze the trends of others physical properties and try to find each
time appropriate reasons for those trends.Periodic trends are specific patterns that are present in the periodic table
that illustrate different aspects of a certain element, including its size and its
electronic properties.Major periodic trends include: atomic radius, electronegativity, ionisation
energy, electron affinity, melting point, density, metallic character.
Periodic trends, arising from the arrangement of the periodic table, provide
chemists with an invaluable tool to quickly predict an element’s properties.
These trends exist because of the similar atomic structure of the elements
within their respective group families or periods, and because of the periodic
nature of the elements.The elements in the periodic table are arranged in order of increasing atomic
number. All of these elements display several other trends and we can
use the periodic table to predict their physical properties. There are many
noticeable patterns in the physical and chemical properties of elements as
we descend in a group or move across a period in the Periodic Table.18.6.1. Atomic radius
The atomic radius is the distance from the centre of the nucleus to the
outermost shell of an atom when it is in the ground state.The covalent radius is one-half the distance between the two nuclei of
identical atoms that are joined together by a single covalent bond.The metallic radius is one-half the distance between the nuclei of two atoms
in contact in the crystalline solid metal.A cation is an atom that has lost one of its outer electrons. Cations have a
smaller radius than the atom that they were formed from.An anion is an atom that has gained an outer electron. Anions have a greater
radius than the atom that they were formed from.Going across a period, the atomic radius decreases. This is caused by the
increase in the number of protons and electrons across a period. One proton
has a greater effect than one electron; thus, electrons are pulled towards the
nucleus, resulting in a smaller radius.Atomic radius increases down the group. This is caused by electron
shielding. The valence electrons occupy higher levels due to the increasing
quantum number. As a result, the valence electrons are further away
from the nucleus as ‘n’ increases. Electron shielding prevents these outer
electrons from being attracted to the nucleus; thus, they are loosely held,
and the resulting atomic radius is large.18.6.2. Electronegativity
Electronegativity is a measure of the relative ability of an atom to attract
the pair of electrons in a covalent bond. Two factors help to determine the
electronegativity value of an element:
• The size of the nuclear charge
• The size of the atomGoing across a period, the electronegativity value increases: as the nuclear
charge increases, the size of the atom decreases and hence there is a greater
attraction between the nucleus and the pair of electrons in a covalent bond.Going down a group, the electronegativity value decreases. The effect of the
increase in the nuclear charge is less than the increase in atomic radius and
the shielding of the inner electrons.Important exceptions of the above rules include the noble gases,
lanthanides and actinides. The noble gases possess a complete valence
shell and do not usually attract electrons. The lanthanides and actinides
possess more complicated chemistry that does not generally follow any
trends. Therefore, noble gases, lanthanides, and actinides do not have
electronegativity values.As for the transition metals, although they have electronegativity values,
there is little variance among them across the period and up and down a
group. This is because their metallic properties affect their ability to attract
electrons as easily as the other elements.According to these two general trends, the most electronegative element is
fluorine, with 3.98 Pauling units.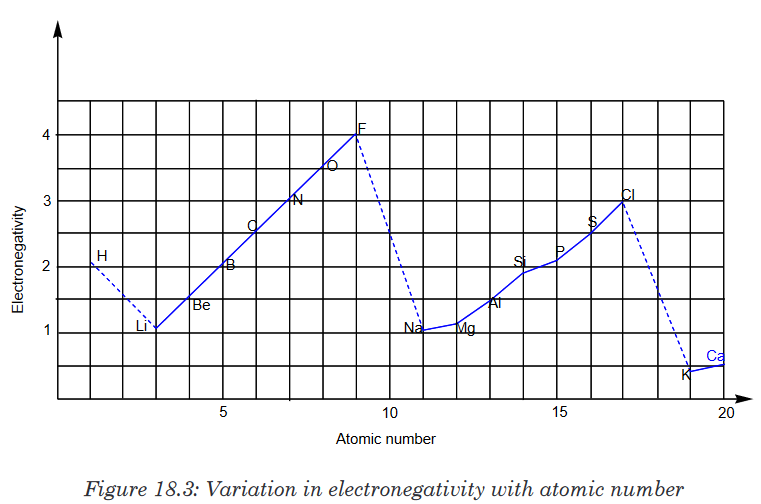 18.6.3.Ionization energy (I.E)
18.6.3.Ionization energy (I.E)
Ionisation energy is the minimum energy (in kJ/mol) required to remove one
mole of electrons from one mole of gaseous atom in the ground state.In general, going across a period, the ionisation energy increases because
the nuclear charge increases and hence electrons are more strongly attracted
to the nucleus.In general, the second ionisation energy of an element is always greater than
the first ionisation. This is explained as follows: every time you remove an
electron from an atom, the remaining electrons are more strongly attracted
by the nucleus and it requires more energy to remove other electrons from
the atom.Hence: 1st IE < 2nd IE < 3rd IEIonisation energy of rare gases or any species with an octet electronic
structure show very high IE because the electron is being removed from a
very stable electronic structure.Down a group, the ionisation energy decreases because the electron which is
removed is further from the nucleus. The nuclear charge also increases, but
the extra inner electrons reduce the effect of the nuclear charge by shielding
the outer electrons from the nucleus.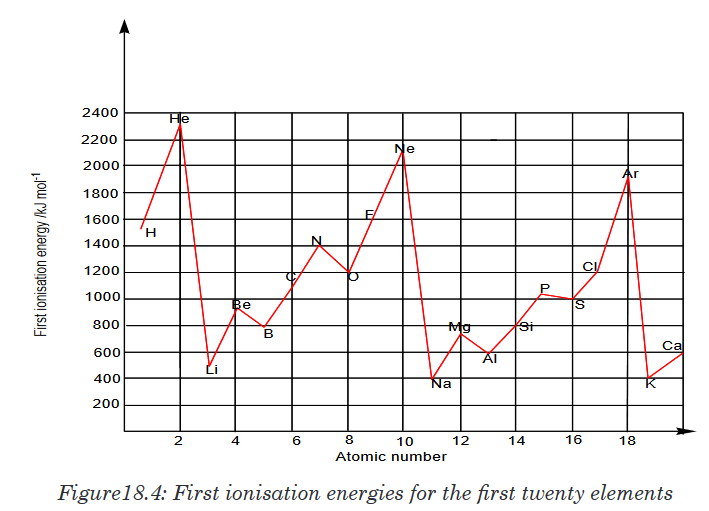 18.6.4. Electron affinity
18.6.4. Electron affinity
As the name suggests, electron affinityis the ability of an atom to accept
an electron. Unlike electronegativity, electron affinity is a quantitative
measurement of the energy change that occurs when an electron is added
to a neutral gas atom. The more negative the electron affinity value, the
higher an atom’s affinity for electrons.That is the electron affinity (E.A.) is the energy change that occurs when an
electron is added to a gaseous atom.It can be either positive or negative value. The greater the negative value,
the more stable the anion is.The electron affinity is positive: X(g)+e−→X−+ Energy (Exothermic)The electron affinity is negative:X(g)+e− + Energy → X− (Endothermic)Electron affinity increases from left to right within a period. This is caused by
the decrease in atomic radius. Moving from left to right across a period, atoms
become smaller as the forces of attraction become stronger. This causes the
electron to move closer to the nucleus, thus increasing the electron affinity
from left to right across a period.Electron affinity decreases from top to bottom within a group. This is
caused by the increase in atomic radius. With a larger distance between the
negatively-charged electron and the positively-charged nucleus, the force of
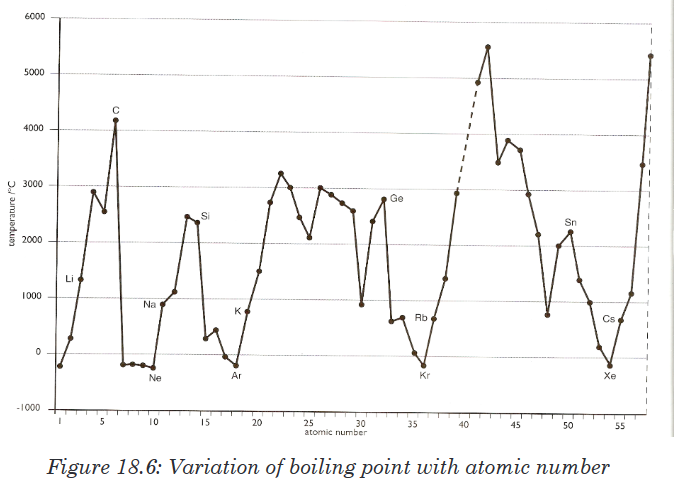
attraction is relatively weaker. Therefore, electron affinity decreases.18.6.5. The melting points and boiling points
Trends can be a bit complicated due to significant structural change from
one element to another in the same group.For groups 1 and 2, the melting and boiling points decrease down the
group. As the atomic radius increases, the strength of the metallic bonding
decreases.For groups 7/17(halogens) and 18/0(noble gases), the melting and boiling
points increase down the group, as the molecule becomes bigger with more
electrons, the Van der Waals forces increase.Across a period, melting and boiling points depend upon the structure and
the bonding in the elements. Sodium, magnesium and aluminium are metals. The strength of the metallic
Sodium, magnesium and aluminium are metals. The strength of the metallic
bonding depends upon the number of delocalised electrons in the metal
structure. The melting point increases from sodium to aluminium.Silicon exists as a giant covalent structure. Each silicon atom is covalently
bonded to four other silicon atoms in a tetrahedral structure (similar to
diamond). A great number of covalent bonds have to be broken to break up
the giant structure.Phosphorus, sulphur and chlorine are all simple molecular species.
Phosphorus consists of P4 molecules; sulphur consists of S8 molecules;
chlorine consists of Cl2 molecules. The strength of the Van der Waals forces
increases as the size of the molecule increases.Argon exists as isolated atoms (it is monatomic) with weak van der Waals
forces between atoms.The illustrations below show the variation of melting and boiling point for
some elements of the periodic table (Figures 18.13 and 18.14).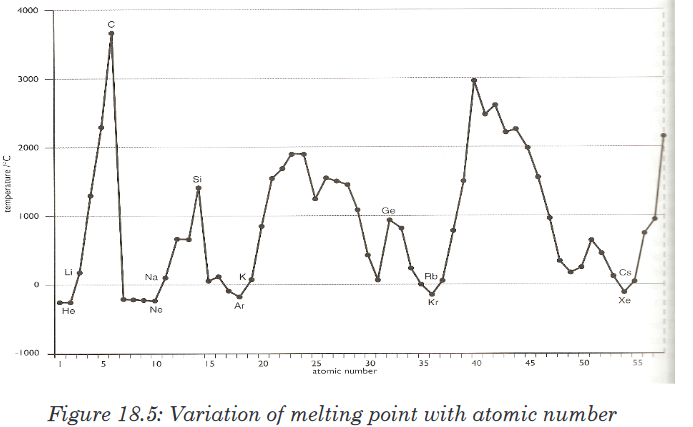
 18.6.6. The density
18.6.6. The density
The density of a substance is its mass per unit volume, usually in g/cm3.
The density is a basic physical property of a homogeneous substance; it
is an intensive property, which means it depends only on the substance’s
composition and does not vary with size or amount.The trends in density of elements can be observed in groups and periods
of the periodic table. In general in any period of the table, the density first
increases from group 1 to a maximum in the centre of the period because themass increases while the size decreases, and then the density decreases
again towards group 18 because of the nature of bonds.Going down a group gives an overall increase in density because even
though the volume increases down the group, the mass increases more.The variation of density with atomic number is shown in the Figure 18.7.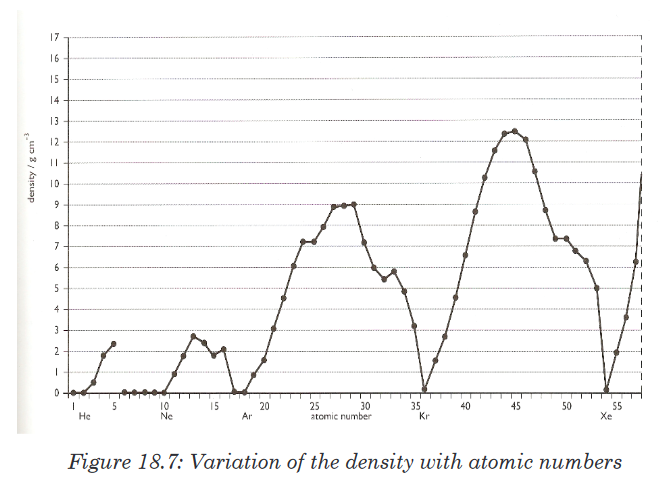 18.6.7. Electrical and thermal conductivity
18.6.7. Electrical and thermal conductivity
The electrical conductivity is the ability of a substance to conduct an electric
current.Across a period, the conductivity increases as the number of delocalised
electrons increases and then decreases as the metallic character decreases
because electronswithin the covalent bonds are held much more tightly in these elements than
in metals.Down a group, the conductivity increases. The delocalised electrons become
less attracted by the nuclei.18.6.8. Electropositivity or Metallic character
Electropositivity or Metallic character refers to the level of reactivity of a
metal. Metals tend to lose electrons in chemical reactions, as indicated by
their low ionization energies.Metals are located in the left and lower three-quarters of the periodic table,
and tend to lose electrons to non-metals. Non-metals are located in the upperright quarter of the table, and tend to gain electrons from metal. Metalloids
are located in the region between the other two classes and have properties.Metallic character is strongest for the elements in the leftmost part of the
periodic table and tends to decrease as we move to the right of any period.Within any group of the representative elements, the metallic character
increases progressively going down.Application activity 18.6
1. Explain why:
a) the atomic radius decreases across a period.
b) electronegativity decreases down a group.
c) Electron affinity increases across a period2. Describe and explain the trend in:
a) Melting point
b) metallic character
i. Across a period (ii) down the group3. Why is it always the case that the 2nd ionisation energy is larger than
the 1st I.E, the 3rd larger than the second I.E and so on?4. In each of the following pairs, indicate which one of the two species is 5. The first and second ionization energies of K are 419 kJ/mol and 3052
5. The first and second ionization energies of K are 419 kJ/mol and 3052
kJ/mol, and those of Ca are590 kJ/mol and 1145 kJ/mol, respectively.
Compare their values and comment on the differencesSkills lab 18
Consider an element with these properties and answer the questions that
follow.• Reacts mildly with 6 M HCl.
• Does not react with base or with water at room temperature.
• Has a good electrical conductivity.
• Is relatively cheap.1. What element has these properties?2. If this element undergoes combustion in air, write a balanced chemical
equation for the reaction.3. How much energy would it take (in kJ) to melt 1 kg of this element?4. Plot a graph of the electrical conductivity for all elements in the group
that includes this element. Is this element the best choice in its group
for an electrical conductor? Explain.5. Would this element make a good copper replacement for wiring in a
house? Explain why or why not.End unit assessment 18
1. What were the limitations of Newland’s classification?
2. The following are coded groups/families of the representative
elements of the periodic table (first 4 periods, s, p blocks only). The
groups are in number of particular order. Use the hints below to
identify the group and place of three elements of each group in their
correct location in the periodic table: AOU, BVW, CKM, DLQ, ENT,
FIJ, GPY, and HRS.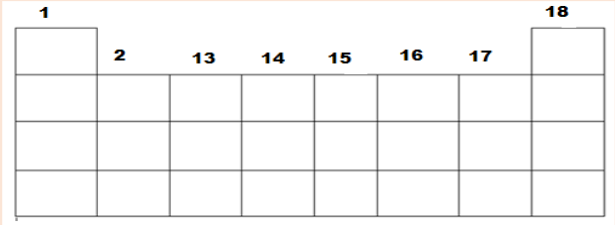 Hints
Hints
A has only one electron in p subshell
B is more electronegative than V
C has a larger atomic radius than both M and W
D has electronic configuration ending in p5
E is one of the most reactive metals
F has a smaller ionization energy than J
G has only 1 energy level with any electrons
H has one more proton than O and is in the same period as OI is the largest alkaline earth metal
J has one more proton than E
K has electron configuration ending in p3
L has more filled energy levels than D
M is larger than K
N has the largest radius in its family
O is smaller than F but in the same energy level as F
P is smaller than Y
Q is the most reactive non-metal
R has the highest electronegativity in its family
T has the lowest density in its family
U more easily loses electrons (think about ionization energy) than
either A or O
V has only 4 electrons in a p-subshell
W has 3 completely filled energy levels
Y has the lowest ionization energy in its family.3. Justify the following statements:
a) The first ionization energy of nitrogen is higher than that of oxygen
even though nuclear charge of nitrogen is less compared to
oxygen.
b) Noble gases are having high ionization energies.4. Explain why:
a) Alkali metals (group 1 elements) are not found free in nature.
b) Atomic radius of gallium is smaller than that of aluminium.(Z of Al
= 13, Z of Ga = 31)5. Arrange these elements in order of decreasing atomic size; sulphur,
chlorine, aluminium and sodium. Does your arrangement demonstrate
a group or a periodic trend?6. List these ions in order of increasing ionic radius: N3-, Na+, F-, Mg2+, O2-
