UNIT 15: STRUCTURE OF AN ATOM AND MASS SPECTRUM
Key Unit competence: Interpret simple mass spectra and use
them to calculate R.A.M. of different
elements.Introductory Activity
The presentations A, B, and C below show three atomic nuclei of different
elements. Study the presentations carefully and answer the questions
below.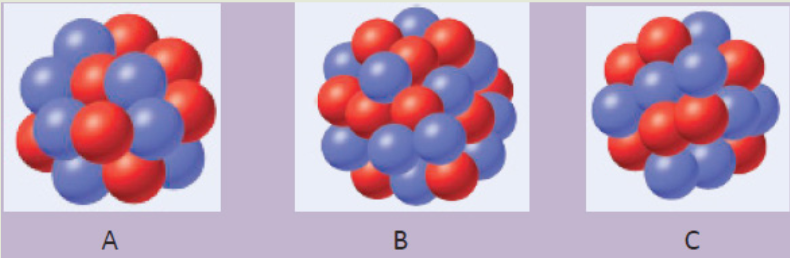
1. How many blue and red spheres do you see in each of the diagrams
above?
2. What do the three diagrams A, B, and C have in common?
3. Based on your knowledge concerning atomic structure, what do you
think that
d) the blue spheres represent?
e) the red spheres represent? Provide explanations.
4.Using the information obtained in question (3) write the atomic symbol
for each of the diagrams.
5.Are there some other particle(s) missing from the above diagrams? If
yes name the particle(s).
6.What could you obtain if the atom is broken down?15.1. The constituents of an atom, their properties and the
outline of their discoveryActivity 15.1
1. Regardless of some exceptions, all atoms are composed of the same
components. True or False? If this statement is true why do different
atoms have different chemical properties?2. The contributions of Joseph John Thomson and Ernest Rutherford
led the way to today’s understanding of the structure of the atom. Use
available resources to figure out:c) the contribution of each of them.
d) the modern view of the structure of the atom?
e) the role of each particle in an atom.Atoms are the basic units of elements and compounds. In ordinary chemical
reactions, atoms retain their identity. An atom is the smallest identifiable unit
of an element. There are about 91 different naturally occurring elements. In
addition, scientists have succeeded in making over 20 synthetic elements
(elements not found in nature but produced in Laboratories of Research
Centres).An element is defined as a substance that cannot be broken down by ordinary
chemical methods in simpler substances. Some examples of elements
include hydrogen (H), helium (He), potassium (K), carbon (C), and mercury
(Hg). In an element, all atoms have the same number of protons or electrons
although the number of neutrons can vary. A substance made of only one
type of atom is also called element or elemental substance, for example:
hydrogen (H2), chlorine (Cl2), sodium (Na). Elements are the basic building
blocks of more complex matter.A compound is a matter or substance formed by the combination of two or
more different elements in fixed ratios. Consider, Hydrogen peroxide (H2O2)
is a compound composed of two elements, hydrogen and oxygen, in a fixed
ratio (2:2).1. Discovery of the atom constituents
The oldest description of matter in science was advanced by the Greek
philosopher Democritus in 400BC.He suggested that matter can be divided into small particles up to an ultimate
particle that cannot any more be divided, and called that particle atom. Atoms
came from the Greek word atomos meaning indivisible.The work of Dalton and other scientists such as Avogadro, etc., contributed
more so that chemistry was beginning to be understood. They proposed new
concept of atom, and from that moment scientists started to think about the
nature of the atom.In 1808 Dalton published A New System of Chemical Philosophy, in which he
presented his theory of atoms:Dalton’s Atomic Theory
a). Each element is made up of tiny particles called atoms.b). The atoms of a given element are identical; the atoms of different
elements are different in some fundamental way(s).c). Chemical compounds are formed when atoms of different elements
combine with each other. A given compound always has the same
relative numbers and types of atoms.d). Chemical reactions involve reorganization of the atoms—changes
in the way they are bound together. The atoms themselves are not
changed in a chemical reaction.
2. Discovery of Electrons and Thomson’s Atomic Model
In 1897 J. J. Thomson (1856–1940) and other scientists conducted several
experiments, and found that atoms are divisible. They conducted experiments
with gas discharge tubes. A gas discharge tube is shown in Figure 15.2.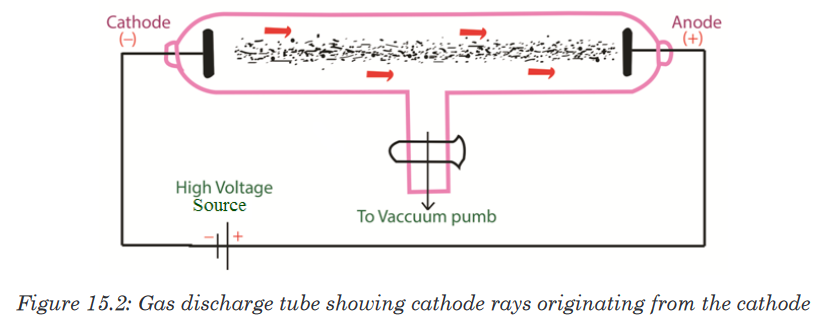
The gas discharge tube is an evacuated glass tube and has two electrodes,
a cathode (negative electrode) and an anode (positive electrode). The
electrodes are connected to a high voltage source. Inside the tube, an
electric discharge occurs between the electrodes.The discharge or ‘rays’ originate from the cathode and move toward the
anode, and hence are called cathode rays. Using luminescent techniques,
the cathode rays are made visible and it was found that these rays are
deflected away from negatively charged plates. The scientist J. J. Thomson
concluded that the cathode rays consist of negatively charged particles, and
he called them electrons.Thomson postulated that an atom consisted of a diffuse cloud of positive
charge with the negative electrons embedded randomly in it. This model,
shown in Figure 15.3, is often called the plum pudding model because the
electrons are like raisins dispersed in a pudding (the positive charge cloud),
as in plum pudding.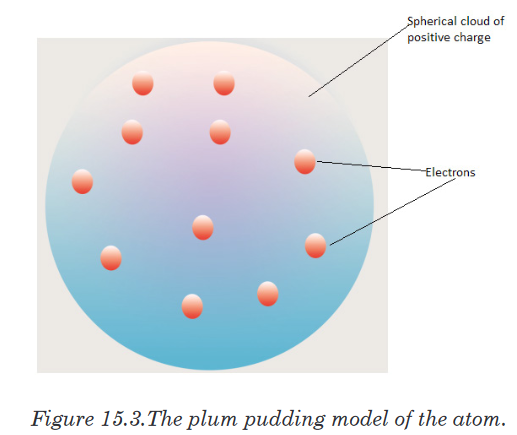
In 1909 Robert Millikan (1868–1953) conducted the famous charged oil
drop experiment and came to several conclusions: He found the magnitude
of the charge of an electron equal to -1.602x10-19C. From the charge-to-
mass ratio(e/m) determined by Thomson, the mass of an electron was also
calculated.
3. Discovery of Protons and Rutherford’s Atomic Model
In 1886 Eugene Goldstein (1850–1930) observed that a cathode-ray tube
also generates a stream of positively charged particles that move towards
the cathode. These were called canal rays because they were observed
occasionally to pass through a channel, or “canal,” drilled in the negative
electrode (Figure 15.4). These positive rays, or positive ions, are created
when the gaseous atoms in the tube lose electrons. Positive ions are formed
by the process.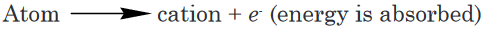
Different elements give positive ions with different e/m ratios. The regularity
of the e/m values for different ions led to the idea that there is a subatomic
particle with one unit of positive charge, called the proton. The proton is a
fundamental particle with a charge equal in magnitude but opposite in sign to
the charge on the electron. Its mass is almost 1836 times that of the electron.
Figure 15.4: A cathode-ray tube with a different design and with a perforated cathode
The proton was observed by Ernest Rutherford and James Chadwick in
1919 as a particle that is emitted by bombardment of certain atoms with
α-particles.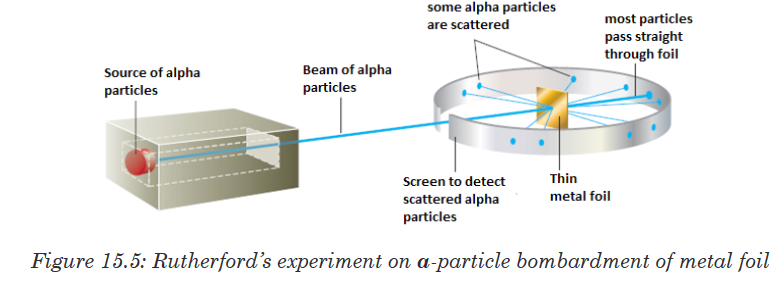
Rutherford reasoned that if Thomson’s model were accurate, the massive
α-particles should crash through the thin foil like cannonballs through gauze,
as shown in Figure 15.6(a). He expected α-particles to travel through the
foil with, at the most, very minor deflections in their paths. The results of the
experiment were very different from those Rutherford anticipated. Although
most of the α- particles passed straight through, many of the particles were
deflected at large angles, as shown in Figure 15.6(b), and some were reflected,
never hitting the detector. This outcome was a great surprise to Rutherford.
Rutherford knew from these results that the plum pudding model for the
atom could not be correct. The large deflections of the α-particles could be
caused only by a centre of concentrated positive charge that contains most
of the atom’s mass, as illustrated in Figure 15.6(b). Most of the α-particles
pass directly through the foil because the atom is mostly empty space. The
deflected α-particles are those that had a “close encounter” with the massive
positive centre of the atom, and the few reflected α-particles are those that
made a “direct hit” on the much more massive positive centre.In Rutherford’s mind these results could be explained only in terms of a
nuclear atom—an atom with a dense centre of positive charge (the nucleus)
with electrons moving around the nucleus at a distance that is large relative
to the nuclear radius.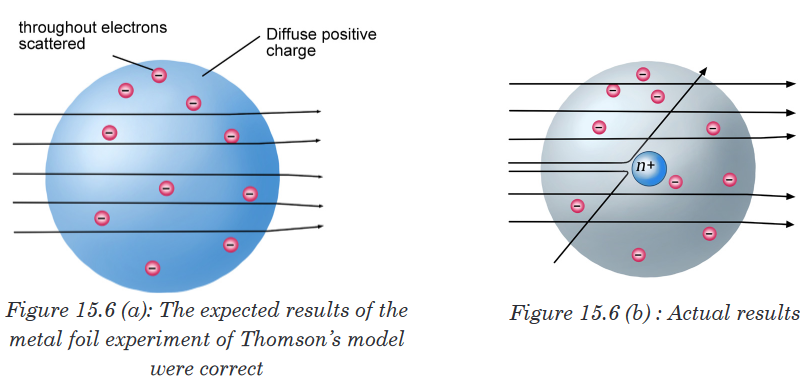
4. Discovery of Neutrons
In spite of the success of Rutherford and his co-workers in explaining atomic
structure, one major problem remained unsolved.If the hydrogen contains one proton and the helium atom contains two
protons, the relative atomic mass of helium should be twice that of hydrogen.
However, the relative atomic mass of helium is four and not two.This question was answered by the discovery of James Chadwick, English
physicist who showed the origin of the extra mass of helium by bombarding
a beryllium foil with alpha particles. (See figure 15.7).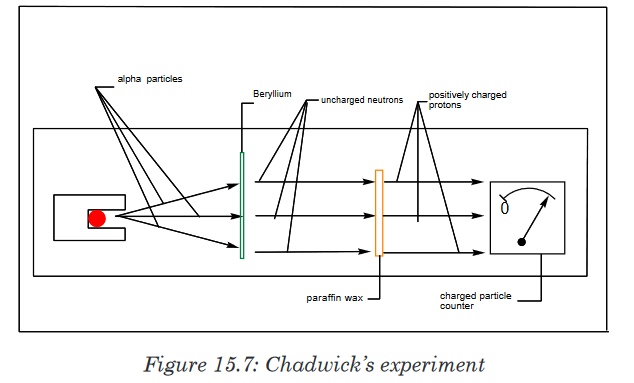
In the presence of beryllium, the alpha particles are not detected; but they
displace uncharged particles from the nuclei of beryllium atoms. These
uncharged particles cannot be detected by a charged counter of particles.However, those uncharged particles can displace positively charged
particles from another substance. They were called neutrons. The mass of
the neutron is slightly greater than that of proton.Figure 15.8 shows the location of the elementary particles (protons, neutrons,
and electrons) in an atom. There are other subatomic particles, but the
electron, the proton, and the neutron are the three fundamental components
of the atom that are important in chemistry.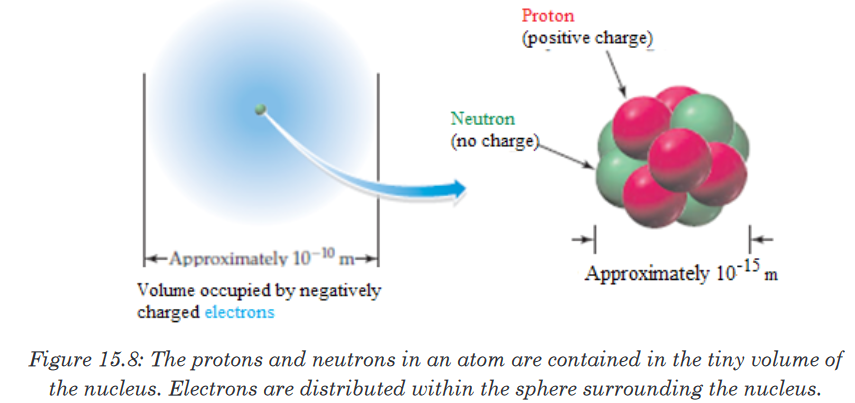
As a result of the experiments described above, it is found that atoms
consist of very small, very dense nuclei surrounded by clouds of electrons at
relatively great distances from the nuclei. All nuclei contain protons; nuclei of
all atoms except the common form of hydrogen also contain neutrons.5. Properties of sub-atomic particles
Protons and neutrons are collectively known as nucleons. Both protons and
neutrons have a mass almost equal to that of hydrogen atom. The neutron
has no charge whereas the proton carries one positive charge. The electron
with one negative charge occupies the space outside the nucleus.The following table summarizes the relative masses, the relative charges
and the position within the atom of these sub-atomic particles.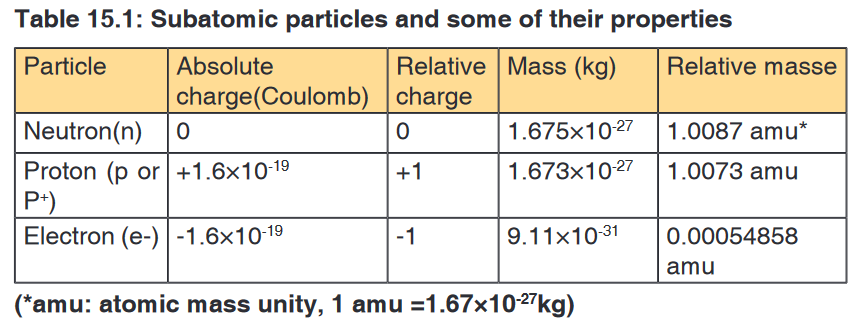
Application activity 15.1
1. In an experiment, it was found that the total charge on an oil drop was
5.93 × 10-18 C. How many negative charges does the drop contain?2. All atoms of the elements contain three fundamental particles. True
or false? Give an example to support your answer.3. Compare the atom constituents
d) in terms of their relative masses
e) in terms of their relative charges4. Using the periodic table as a guide, specify the number of protons and
electrons in a neutral atom of each of these elements.
a). Carbon (C) b). Calcium (Ca) c).Chlorine (Cl) d). Chromium (Cr)15.2. Concept of atomic number, mass number, isotopic
mass and relative atomic mass
Activity 15.2
Atoms of the same element may exhibit some physical properties like
mass, density, velocity. Is the statement true or false? Use available
textbooks and suggest an appropriate explanation?The atomic number or proton number, Z, denotes the number of protons in
an atom’s nucleus. It corresponds to the order of the element in the periodic
table.The mass number or nucleon number, A, denotes the total number of protons
and neutrons in an atom.Mass number = number of protons + number of neutrons
= atomic number + neutron number
The number of neutrons can be obtained by subtracting the atomic number
from the mass number.Chemists use the following shorthand to represent an atom. The mass
number is shown as a superscript (top number) and the atomic number is
shown as a subscript (bottom number) beside the symbol of the element.By convention, the atomic number is usually written to the left subscript of
the elemental notation, and the mass number to the left superscript of the
elemental notation as represented by the example below, where X represents
any element symbol.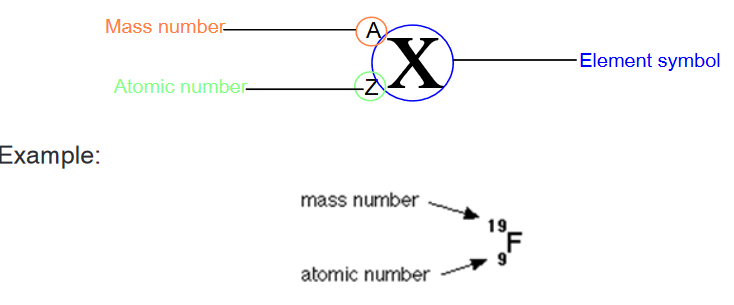
Some atoms of the same element have the same atomic number, but different
mass numbers. This means a different number of neutrons. Such atoms are
called isotopes of the element. They are nuclides of the same element.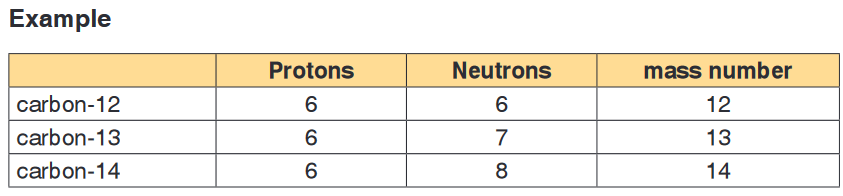
Isotopes of an element have the same chemical properties because they
have the same number of electrons.When elements react, it is the electrons that are involved in the reactions.
This means that the isotopes of an element cannot be differentiated by
chemical reactions.Because isotopes of an element have different numbers of neutrons,
they have different masses, and isotopes have slightly different physical
properties.The mass of a single isotope is its isotopic mass. The relative isotopic mass
of an isotope is the relative mass of that isotope compared with the isotope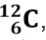 which is given a mass of 12.00 units (12.00 atomic mass units). That is,
which is given a mass of 12.00 units (12.00 atomic mass units). That is,
relative isotopic mass relates to the relative atomic mass scale on which one
isotope of the carbon element, carbon-12 is taken as the reference standard
for atomic masses and is given a relative mass of 12 units, precisely 12
atomic mass units (a.m.u).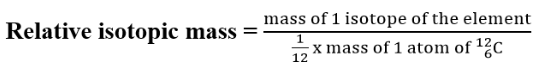
Atomic mass unit (a.m.u) is a unit of mass used to express “relative atomic
masses”. It is 1/12 of the mass of the isotope of carbon-12 and is equal to
1.66054x10-27kg.The relative isotopic masses of all other atoms are obtained by comparison
with the mass of a carbon-12 atom.On that scale, the relative atomic mass of a proton and that of a neutron are
both very close to one unit (1.0074 and 1.0089 units respectively). Since
the relative mass of an electron is negligible (0.0005units), it follows that all
isotopic masses will be close to whole numbers.However relative atomic masses of elements are not close to whole numbers
because natural occurring elements are often mixtures of isotopes.Application activity 15.2
1. How do you call the members of each of the following pairs? Explain.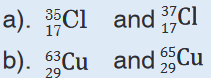
2. Write, using the periodic table, the correct symbols to identify an atom
that contains
a). 4 protons, 4 electrons, and 5 neutrons;
b). 23 protons, 23 electrons, and 28 neutrons;
c). 54 protons, 54 electrons, and 70 neutrons; and
d). 31 protons, 31 electrons, and 38 neutrons.3. Use the list of the words given below to fill in the blank spaces. Each
word will be used once.
Atomic number, Mass number, protons, Electrons, Isotopes, neutron
a). The atomic number tells you how many ___________________
and ____________________ are in an atom.
b). ____________________ is the number written as subscript on
the left of the atomic symbol.
c). The total number of protons and neutrons in an atom is called
the ____________.
d). Atoms with the same number of protons but different number of
neutrons are called ________________.
e). The subatomic particle that has no charge is called a15.3. Calculations of the relative atomic masses of elements
Activity 15.3
Using textbooks and internet connection, explain the concept of relative
mass and attempt to solve the problems below.
1. Argon has three naturally occurring isotopes: argon-36, argon-38,
and argon-40. Based on argon’s reported relative atomic mass from
the periodic table, which isotope do you think is the most abundant in
nature? Explain
The Relative atomic mass, symbolized as R.A.M (Ar), is defined as the
average of the relative isotopic masses of the different isotopes weighted in
the proportions in which they naturally occur.Thus, the different isotopic masses of the same elements and the percentage
abundance of each isotope of an element must be known in order to
accurately calculate the relative atomic mass of an element.Notice: Remember that mass number is not the same as the relative atomic
mass or isotopic mass! The mass number is the total number of protons and
neutrons while relative atomic mass is the average of the isotopic masses.
By applying the same formula, the relative abundance of the isotopes may be
calculated knowing the relative atomic mass of the element and the atomic
masses of the respective isotopes.
Application activity 15.3
1. Three isotopes of magnesium occur in nature. Their abundances and
masses, determined by mass spectrometry, are listed in the following
table. Use this information to calculate the relative atomic mass of
magnesium.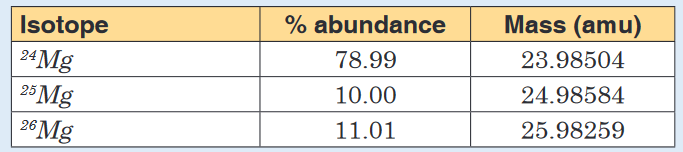
2. The atomic weight of gallium is 69.72 amu. The masses of the
naturally occurring Isotopes are 68.9257 amu for 69Ga and 70.9249
amu for 71Ga. Calculate the percent abundance of each isotope.3. Natural occurring lead contains 1.55% lead-204, 23.6% lead-206,
22.6% lead-205 and 52.3% lead -208. Calculate the relative atomic
mass of lead.4. The table below shows the mass number and the percentage
abundances of the isotopes of an element X
e) Calculate the relative atomic mass of X
f) Identify the element X.
15.4. Mass spectrometer15.4.1. Description and functioning of its components
Activity 15.4.1
1. Recall the concept of relative atomic mass of an element.
2. Do research and identify the name of an instrument used to
estimate the mass and proportions of the isotopes that make up
a given element. How does it work?The mass spectrometer is an instrument that separates positive gaseous
atoms and molecules according to their mass-charge ratio and that records
the resulting mass spectrum.In the mass spectrometer, atoms and molecules are converted into ions. The
ions are separated as a result of the deflection which occurs in a magnetic
and electric field.The basic components of a mass spectrometer are: vaporisation chamber
(to produce gaseous atoms or molecules), ionization chamber (to produce
positive ions), accelerating chamber (to accelerate the positive ions to a high
and constant velocity), magnetic field (to separate positive ions of different
m/z ratios), detector (to detect the number and m/z ratio of the positive ions)
and the recorder (to plot the mass spectrum of the sample).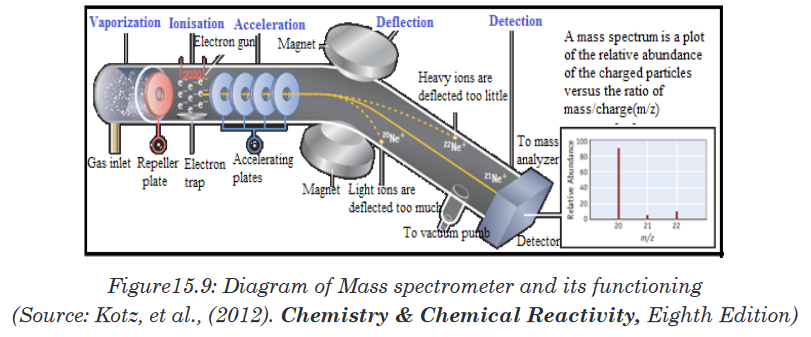
A mass spectrometer works in five main stages, namely vaporization,
ionization, acceleration, deflection, and detection to produce the mass
spectrum.Stage 1: Vaporization
At the beginning, the test sample is heated until it becomes vapour and is
introduced as a vapour into the ionization chamber. When a sample is a solid
with low vapour pressure, it can directly be introduced into the ionization
chamber.Stage 2: Ionisation
The vaporized sample passes into the ionization chamber (with a positive
voltage of about 10,000 volts). The electrically heated metal coil gives
off electrons which are attracted to the electron trap which is a positively
charged plate.The particles in the sample (atoms or molecules) are therefore bombarded
with a stream of electrons (electrons gun), and some of the collisions are
energetic enough to knock one or more electrons out of the sample particles
to make positive ions. Mass spectrometers always work with positive ions.Most of the positive ions formed will carry a charge of +1 because it is much
more difficult to remove further electrons from an already positive ion.Most of the sample molecules are not ionized at all but are continuously
drawn off by vacuum pumps which are connected to the ionization chamber.
Some of the molecules are converted to negative ions through the absorption
of electrons.Stage 3: Acceleration
The positive ions are accelerated by an electric field so that they move
rapidly through the machine at high and constant velocity.Stage 4: Deflection
The ions are then deflected by a magnetic field according to their masses
and charges ratio. Different ions are deflected by the magnetic field at
different extents. The extent to which the beam of ions is deflected depends
on four factors:1. The magnitude of the accelerating voltage (electric field strength).
Higher voltages result in beams of more rapidly moving particles to
be deflected less than the beams of the more slowly moving particles
produced by lower voltages.2. Magnetic field strength. Stronger fields deflect a given beam more
than weaker fields.3. Masses of the particles. Because of their inertia, heavier particles
are deflected less than lighter particles that carry the same charge.4. Charges on the particles. Particles with higher charges interact
more strongly with magnetic fields and are thus deflected more than
particles of equal mass with smaller chargesThe two last factors (mass of the ion and charge on the ion) are combined
into the mass/charge ratio. Mass/charge ratio is given the symbol m/z (or
sometimes m/e).For example, if an ion had a mass of 28 and a charge of 1+, its mass/charge
ratio would be 28. An ion with a mass of 56 and a charge of 2+ would also
have a mass/charge ratio of 28.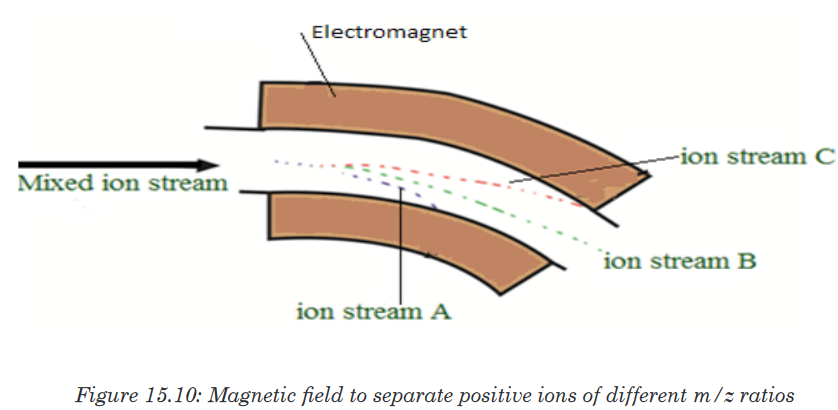
In the figure 15.10 above, ion stream A is most deflected: it will contain ions
with the smallest mass/charge ratio. Ion stream C is the least deflected: it
contains ions with the greatest mass/charge ratio. Assuming 1+ ions, stream
A has the lightest ions, stream B the next lightest and stream C the heaviest.
Lighter ions are going to be more deflected than heavy ones.Stage 5: Detection
The beam of ions passing through the machine is detected electrically. As
they pass out of the magnetic field, ions are detected by an ion detector,
which records the position of the ions on the screen and the number of ions
that hit the screen at each position. These two pieces of information are
used to produce a mass spectrum for the sample.A flow of electrons in the wire is detected as an electric current, which can
be amplified and recorded. The more ions arriving, the greater the current.Detecting the other ions
How might the other ions be detected (those in streams A and C which have
been lost in the machine)?Remember that stream A was most deflected. To bring them on to the
detector, you would need to deflect them less by using a smaller magnetic
field.To bring those with a larger m/z value (the heavier ions if the charge is +1) to
the detector you would have to deflect them more by using a larger magnetic
field.If you vary the magnetic field, you can bring each ion stream in turn on
the detector to produce a current which is proportional to the number of
ions arriving. The mass of each ion being detected is related to the size
of the magnetic field used to bring it on to the detector. The machine can
be calibrated to record current (which is a measure of the number of ions)
against m/z directly. The mass is measured on the 12C scale.Note: The 12C scale is a scale on which the 12C isotope weighs exactly 12
units.Recorder
The detector of a typical instrument consists of a counter, which produces
a current that is proportional to the number of ions, which strike it. Using of
electron multiplier circuits, this current can be measured so accurately that
the current caused by just one ion striking the detector can be measured.
The signal from the detector is fed to a recorder, which produces the mass
spectrum. In modern instruments, the output of the detector is fed through
an interface to a computer. The computer can store the data, provide the
output in both tabular and graphic forms, and compare the data to standard
spectra, which are contained in spectra libraries also stored in the computer.The following figure is an example of a mass spectrum of unknown element
that has 2 isotopes.
Application activity 15.4.1
1. Use the list of the words given below to fill in the blank spaces. Each
word will be used once.Vaporization chamber, mass spectrum, velocity, ionization, deflection,
detector, accelerationA sample of the element is placed in the _________ chamber where
it is converted into gaseous atoms. The gaseous atoms are ionized
by bombardment of high energy electrons emitted by a hot cathode
to become positive ions (in practice, the voltage in the ________
chamber is set in such a way that only one electron is removed from
each atom). The positive ions (with different masses) are then going
faster to a high and constant _________by two negatively charged
plates: the process is called_________. The positive ions are then
deviated by the magnet field. This process is called ____________
(ions with smaller mass will be deflected more than the heavier ones).
These ions are then detected by the ion _________. The information
is fed into a computer which prints out the________ of the element.2. The correct order for the basic features of a mass spectrometer is...
a) acceleration, deflection, detection, ionization
b) ionisation, acceleration, deflection, detection
c) acceleration, ionisation, deflection, detection
d) acceleration, deflection, ionisation, detection3. Which one of the following statements about ionisation in a mass
spectrometer is incorrect?a) gaseous atoms are ionised by bombarding them with high energy
electronsb) atoms are ionised so they can be accelerated
c) atoms are ionised so they can be deflected
d) it doesn’t matter how much energy you use to ionise the atoms
4. The path of ions after deflection depends on...
a) only the mass of the ion
b) only the charge on the ion
c) both the charge and the mass of the ion
d) neither the charge nor the mass of the ion5. Which of the following species will be deflected to the greatest extent?
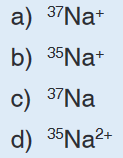
6. Which of the following separates the ions according to their mass-to-
charge?
a) Ion source
b) Detector
c) Magnetic sector
d) Electric sector15.4.2. Interpretation of mass spectra and uses of the mass
spectrometer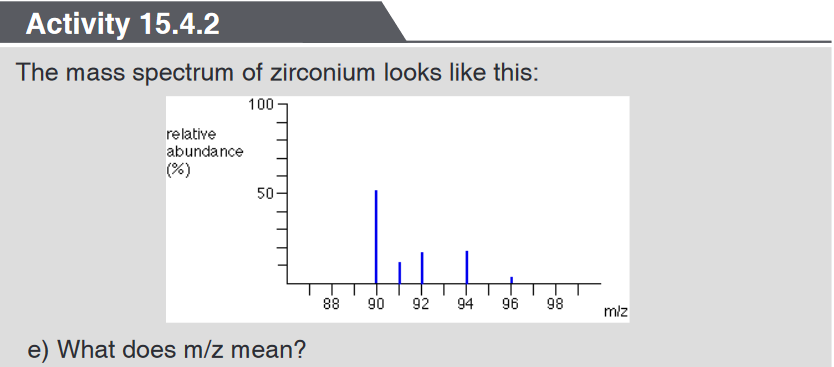
f) Explain as fully as possible what the mass spectrum shows about
zirconium. (You are not required to read actual values from the relative
abundance axis.)g) The spectrum shows lines for 1+ ions. If there were also peaks for 2+
ions, where would you expect to find them, and what would you predict
about their heights relative to the 1+ peaks?The mass spectrum of an element shows how you can find out the masses
and relative abundances of the various isotopes of the element and use that
information to calculate the relative atomic mass of the element.Example 1: The mass spectrum of boron

The mass spectrum of boron may be used to know the number of boron
isotopes and their relative abundances.The two peaks in the mass spectrum shows that there are 2 isotopes of
boron with relative isotopic masses of 10 and 11 on the 12C scale.The relative size of the peaks gives you a direct measure of the relative
abundances of the isotopes. The tallest peak is often given an arbitrary
height of 100 but you may find all sorts of other scales used; it doesn’t
matter. You can find the relative abundances by measuring the lines on the
stick diagram.In this case, the two isotopes (with their relative abundances) are:


The mass spectrum of zirconium may be used to know the number of
zirconium isotopes and their relative abundancesThe five peaks in the mass spectrum show that there are five isotopes of
zirconium with relative isotopic masses of 90, 91, 92, 94 and 96 on the 12C
scale.This time, the relative abundances are given as percentages. Again you can
find these relative abundances by measuring the lines on the stick diagram.
In this case, the five isotopes (with their relative percentage abundances)
are:
Example 3: The mass spectrum of chlorine
Chlorine is taken as typical of elements with more than one atom per molecule.
Chlorine has two isotopes, 35Cl and 37Cl, in the approximate ratio of 3 atoms
of 35Cl to 1 atom of 37Cl. You might suppose that the mass spectrum would
look like this: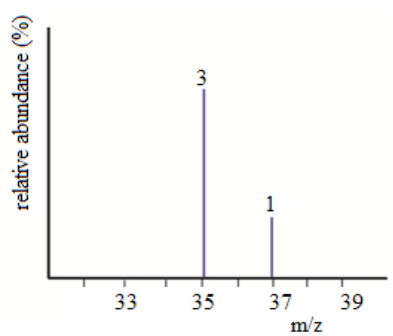
But it is not true. The problem is that chlorine consists of molecules, not
individual atoms. When chlorine is passed into the ionization chamber, an
electron is knocked off the molecule to give a molecular ion, Cl2+. These
ions won’t be particularly stable, and some will fall apart to give a chlorine
atom and a Cl+ ion. The term for this is fragmentation.
If the Cl atom formed isn’t then ionized in the ionization chamber, it simply
gets lost in the machine (neither accelerated nor deflected).The Cl+ ions will pass through the machine and will give lines at 35 and 37,
depending on the isotope and you would get exactly the pattern in the last
diagram. The problem is that you will also record lines for the unfragmented
Cl2+ ions.At the end the spectrum will show peaks due to ionized atoms, Cl+ at 35, and
37, and ionized molecule Cl2
+ at 70, 72, 74 as in the figure 15.12 below.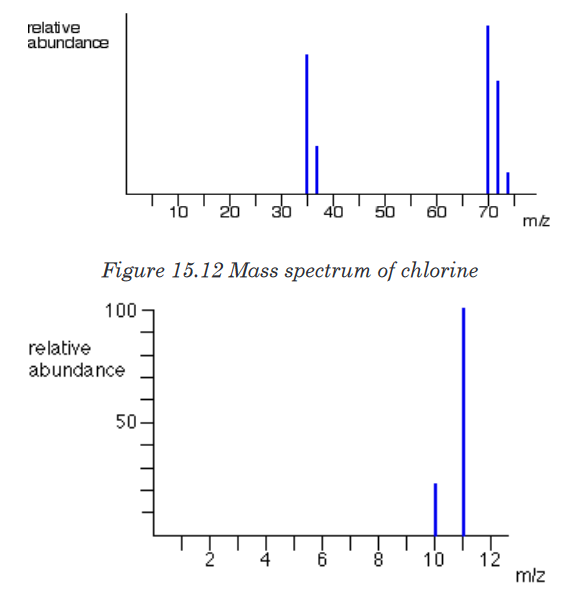
Relative atomic masses can be calculated using the information from the
mass spectrum.Example 1: The mass spectrum of boron is given below:
Determine the relative atomic mass of boron
From the mass spectrum given, we have123 typical atoms of boron (sum of
relative abundances). 23 of these would be 10B and 100 would be 11B.The total mass of these would be (23 x 10) + (100 x 11) = 1330
The average mass of these 123 atoms would be 1330 / 123 = 10.8 (to 3
significant figures).10.8 is the relative atomic mass of boron.
Example 2: The figure below represents the mass spectrum of zirconium
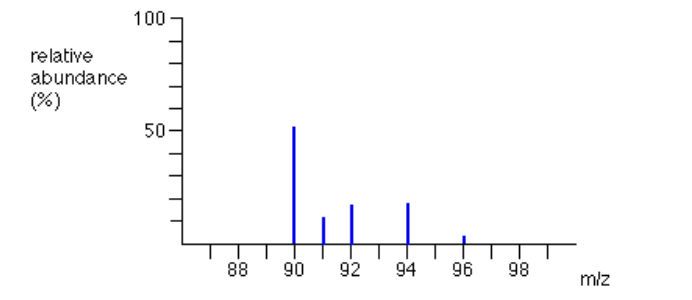
Suppose you had 100 typical atoms of zirconium (sum of relative abundances).
51.5 of these would be 90Zr, 11.2 would be 91Zr and so on. The total mass of
these 100 typical atoms would be(51.5 x 90) + (11.2 x 91) + (17.1 x 92) + (17.4 x 94) + (2.8 x 96) = 9131.8
The weighted average mass of these 100 atoms would be 9131.8 / 100 =
91.3 (to 3 significant figures).91.3 is the relative atomic mass of zirconium.
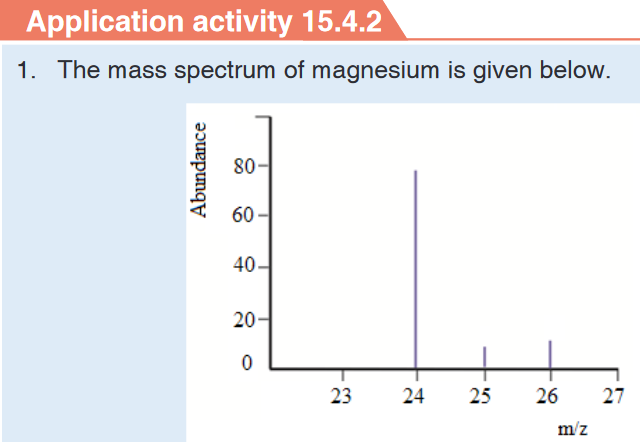
b) How many isotopes does magnesium possess
c) Estimate the isotopic mass of each of the magnesium isotopes
d) Estimate the relative abundance for each of the isotopes of
magnesium2. Which of the following is not done through mass spectrometry?
e) Calculating the isotopic abundance of elements
f) Investigating the elemental composition of planets
g) Confirming the presence of O-H and C=O in organic compounds
h) Calculating the molecular mass of organic compounds3. When a pure, gaseous sample of element X is introduced in a mass
spectrometer, four mononuclear, singly charged ions are detected as
shown in the spectrum below.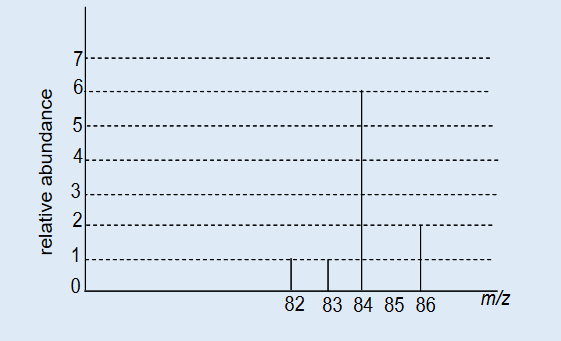
i) Describe the process by which the gaseous sample of X is converted
into ions in a mass spectrometer.j) What adjustment is made to the operating system in order to direct
the different ions, in turn, onto, the detector of a mass spectrometer?k) What is the number of isotopes of element X? Justify your answer.
l) Use the data from the spectrum above to calculate the relative atomic
mass of X.m)Identify the element X.
Skills lab 15
When atoms of any element are subjected to high voltages, cathode rays
are emitted and are the same for all elements.During your free time, write an essay about all other radiations that may be
emitted by atoms specifying the conditions under which they can be emitted
and their properties. In your essay, you have to point out the benefits and
the disadvantages of those radiations to humans and their environment.End unit assessment 15
I. Multiple choice questions
1. Which of the following is true regarding a typical atom?
a) Neutrons and electrons have the same mass.
b) The mass of neutrons is much less than that of electrons.
c) Neutrons and protons together make the nucleus electrically
neutral.
d) Protons are more massive than electrons2. Which of the following statements is(are) true? For the false
statements, correct them.
a) All particles in the nucleus of an atom are charged.
b) The atom is best described as a uniform sphere of matter in which
electrons are embedded.
c) The mass of the nucleus is only a very small fraction of the mass
of the entire atom.
d) The volume of the nucleus is only a very small fraction of the total
volume of the atom.
e) The number of neutrons in a neutral atom must equal the number
of electrons.3. Each of the following statements is true, but Dalton might have
had trouble explaining some of them with his atomic theory. Give
explanations for the following statements.
a) Atoms can be broken down into smaller particles.
b) One sample of lithium hydride is 87.4% lithium by mass, while
another sample of lithium hydride is 74.9% lithium by mass.
However, the two samples have the same chemical properties4. In mass spectrometer, the sample that has to be analyzed is
bombarded with which of the following?
a) Protons
b) Electrons
c) Neutrons
d) Alpha particles5. Mass spectrometer separates ions on the basis of which of the
following?
a) Mass
b) Charge
c) Molecular weight
d) Mass to charge ratio6. In a mass spectrometer, the ions are sorted out in which of the following
ways?
a) By accelerating them through electric field.
b) By accelerating them through magnetic field.
c) By accelerating them through electric and magnetic field.
d) By applying a high voltage.7. The procedure for mass spectroscopy starts with which of the following
processes?
a) The sample is bombarded by electron beam.
b) The ions are separated by passing them into electric and magnetic
field.
c) The sample is converted into gaseous state.
d) The ions are detected.8. Which of the following ions pass through the slit and reach the collecting
plate?
a) Negative ions of all masses.
b) Positive ions of all masses.
c) Negative ions of specific mass.
d) Positive ions of specific mass.9. Which of the following statements is not true about mass spectrometry?
a) Impurities of masses different from the one being analyzed
interferes with the result.
b) It has great sensitivity.
c) It is suitable for data storage.
d) It is suitable for library retrieval.10. In a mass spectrometer, the sample gas is introduced into the highly
evacuated spectrometer tube and it is ionised by the electron beam.
a) True
b) FalseII. Short and long answer questions
11. What are the three fundamental particles from which atoms are built?
What are their electric charges? Which of these particles constitute
the nucleus of an atom? Which is the least massive particle of the
fundamental particles?12. Verify that the atomic weight of lithium is 6.94, given the following
information:
6Li, mass = 6.015121 u; percent abundance = 7.50%
7Li, mass = 7.016003 u; percent abundance = 92.50%13. The diagram below shows the main parts of a mass spectrometer.
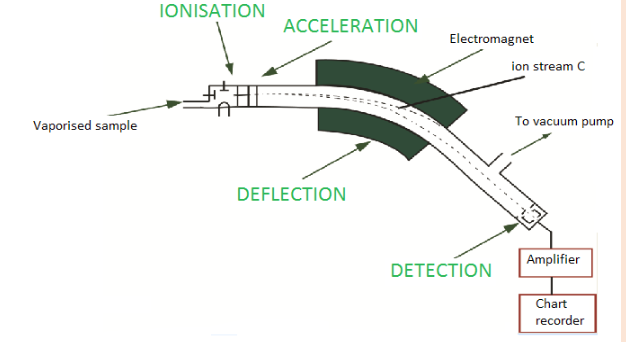
a) Describe the different steps involved in taking a mass spectrum of a
sample
i. Which two properties of the ions determine how much they are
deflected by the magnetic field? What effect does each of these
properties have on the extent of deflection?
ii. Of the three different ion streams in the diagram above, why is
the ion stream C least deflected?
iii. What would you have to do to focus the ion stream C on the
detector?b) Why is it important that there is a vacuum in the instrument?
c) Describe briefly how the detector works.
14.
(a) A mass spectrum of a sample of indium shows two peaks at m/z
= 113 and m/z = 115. The relative atomic mass of indium is 114.5.
Calculate the relative abundances of these two isotopes.(b)The mass spectrum of the sample of magnesium contains three
peaks with the mass-to-charge rations and relative intensities
shown below
i. Explain why magnesium gives three peaks in mass spectrum?
ii. Use the information in the table above to calculate the accurate
value for the relative atomic mass of magnesium15. There exists 3 isotopes of oxygen that occur naturally with atomic mass
16, 17 and 18 with abundance 99.1% ; 0.89% and 0.01% respectively.
Given that oxygen occurs naturally as diatomic molecule,
a) Predict the number of peaks that will be observed on the screen
of mass spectrometer.
b) Show the molecular ions that are responsible of these peaks.
