UNIT 1: PRINCIPLES OF PHARMACOLOGY
Key Unit Competence
Apply fundamental principles of pharmacology during patient care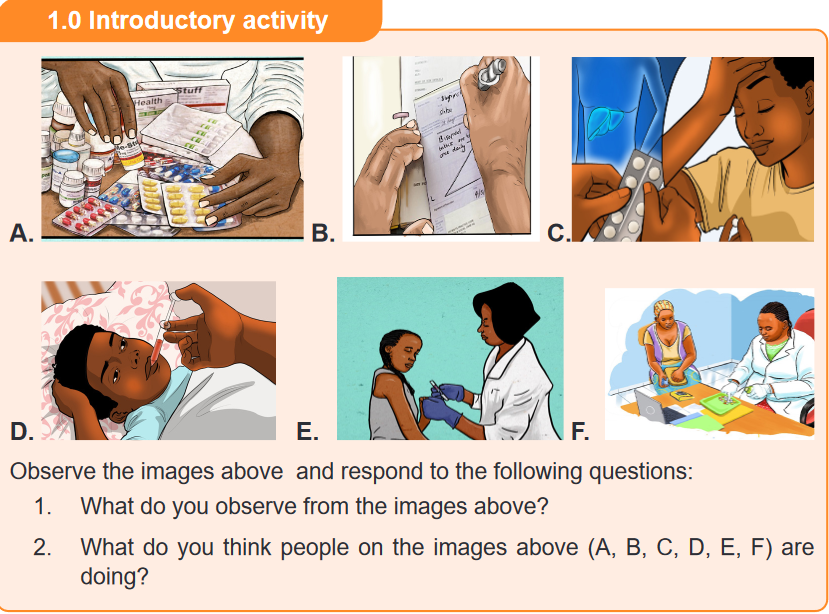
1.1. History of pharmacology
Learning activity 1.1
Read the case study below and answer the questions related to it:
A student in O’ level is concerned about different ways of managing illness.
Therefore, he asked different ways, sources and reasons of using medications.
As a student who has chosen the associate nursing program, you know that a
medicine is linked to the pharmacology science and you expect to use medicines
to help persons who have diseases.
1. What do you think are the sources of medicines?
2. What is the purpose of using drug substances in human kind?CONTENT SUMMARY
The story of pharmacology is rich and exciting, filled with accidental or unplanned
discoveries and landmark events. Its history likely began when a human first used
a plant to relieve symptoms of disease. One of the oldest forms of health care,
herbal medicine has been practiced in virtually every culture dating to antiquity /
ancient times.The Babylonians recorded the earliest surviving prescriptions on clay tablets
in 3000 Before Christ (BC), although magic and the art of reading omens were
probably considered as legitimate to healing as the use of drug remedies.At about the same time, the Chinese recorded the Pen Tsao (Great Herbal), a
40-volume compendium of plant remedies dating to 2700 BC. The Egyptians
followed in 1500 BC by archiving their remedies on a document known as the Eber
s papyrus, which contains over 700 magical formulas and remedies. Galen, the
famous Greek physician, described over 1,000 healing preparations using plant
products before his death in Dark Ages (AD) 201.Pharmacology as a distinct discipline was officially recognized when the first
Department of Pharmacology was established in Estonia in 1847. John Jacob
Abel, was considered as the father of American pharmacology due to his many
contributions to the field, founded the first pharmacology department in the United
States at the University of Michigan in 1890.Drugs are substances that are used in the diagnosis, prevention, treatment or
cure of diseases. In early times, these substances were derived from natural
sources, of which plants took up the major share. With the introduction of technology,
most drugs today are manufactured synthetically in the laboratory.The major sources of drugs can be grouped into the following: Plant, animal, mineral,
marine, synthetic/chemical derivative, Semi-synthetic, Microbiological and
Recombinant DNA technology/ Biosynthetic sources.1. Plant source
It is the oldest source of drugs. Most of the drugs in ancient times were derived
from plants. A number of plants have medicinal qualities and have been used for
centuries as drugs or drug sources. Although the earliest plant source for drugs
was the leaf, other parts of plants (e.g., barks, fruits, roots, stem, wood, seeds,
blossoms, bulb etc.) Almost all parts of the plants are used i.e. leaves, stem, bark,
fruits and roots.Leaves: The leaves of Digitalis Purpurea are the source of Digitoxin and Digoxin,
which are cardiac glycosides; used to treat HF (heart failure).
Leaves of Eucalyptus give oil of Eucalyptus, which is important component of cold
& cough syrup.
Flowers: Poppy papaver somniferous gives morphine (opoid), Vinca rosea gives
vincristine and vinblastine and Rose gives rose water used as tonic.Fruits: Senna pod gives anthracine, which is a purgative and Calabar beans Give
physostigmine, which is cholinomimetic agent.Seeds: Seeds of Nux Vomica give strychnine, which is a CNS stimulant and Castor
seeds give castor oil.Roots: Ipecacuanha root gives Emetine, used to induce vomiting as in accidental
poisoning, it also has amoebicidal properties.
Ipecacuanha root gives Emetine, used to induce vomiting as in accidental poising,
it also has ameobicidal properties.Rauwolfia serpentina gives reserpine, a hypotensive agent. Reserpine was used
for hypertension treatment.
Bark: Cinchona bark gives quinine and quinidine, which are antimalarial drugs.
Quinidine also has antiarrhythmic properties.
Cinchona hark gives quinine and quinidine, which are antimalarial drugs
Atropa belladonna gives atropine, which is anticholinergic. Hyoscyamus Niger
gives Hyosine, which is also anticholinergic.
Stem: Chondrodendron tomentosum gives tuboqurarine, which is skeletal muscle
relaxant used in general anaesthesia.2. Animal sources
Many important drugs are derived from animal source. In most instances, these
medicinal substances are derived from the animal’s body secretions, fluid or glands.
Insulin, heparin, adrenaline, thyroxin, cod liver oil, musk, beeswax, enzymes, and
antitoxins sera are some examples of drugs obtained from animal sources. Like
plant products, drugs from animal sources may be crude (unrefined) or refined
material.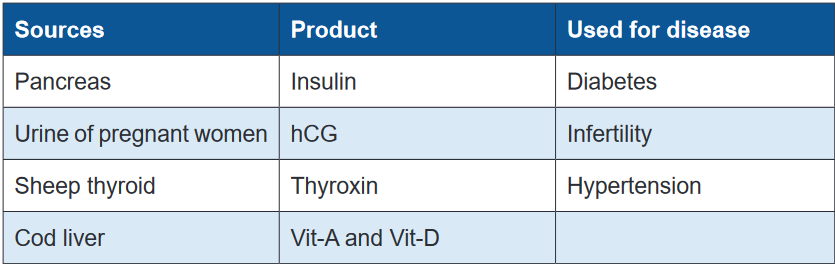
3. Mineral sources
Minerals (both metallic and non-metallic minerals) have been used as drugs
since ancient times. Our body requires trace elements of minerals in order to
maintain homeostasis. Patients lacking an adequate level of these materials may
take specific mineral-based drugs to raise the level of minerals.Examples include ferrous sulfate in iron deficiency anemia; magnesium sulfate as
purgative; magnesium trisilicate, aluminum hydroxide and sodium bicarbonate as
antacids for hyperacidity and peptic ulcer; zinc oxide ointment as skin protectant,
in wounds and eczema; gold salts as anti-inflammatory and in rheumatoid arthritis;
selenium as anti-dandruff.• Metallic and non-metallic sources: Iron is used in treatment of iron
deficiency anemia, Mercurial salts are used in Syphilis (bacterial infection),
Zinc is used as zinc supplement, Zinc oxide paste is used in wounds and in
eczema, Iodine is antiseptic and Iodine supplements are also used and Gold
salts are used in the treatment of rheumatoid arthritis• Miscellaneous sources: Fluorine has antiseptic properties, Borax has
antiseptic properties as well, Selenium as selenium sulphide is used in
antidandruff shampoos and Petroleum is used in preparation of liquid paraffin.4. Marine source (water source)
Bioactive compounds from marine flora and fauna have extensive past and
present use in the prevention, treatment or cure of many diseases. Fish andmarine microorganisms produce biologically potent chemicals with interesting anti-
inflammatory, anti-viral, and anticancer activity.5. Synthetic /chemical derivativeA synthetic drug is produced using chemical synthesis, which rearranges chemical
derivatives to form a new compound. The synthetic sources of drugs evolved with
human skills in the laboratory and advanced knowledge and understanding of
phytochemical investigation. When the nucleus of the drug from natural source
as well as its chemical structure is altered, we call it synthetic. Examples include
Emetine Bismuth Iodide. At present, majority of drugs used in clinical practice are
exclusively prepared synthetically in pharmaceutical and chemical laboratory.6. Semi-synthetic SourcesSemi-synthetic drugs are neither completely natural nor completely synthetic. They
are a hybrid and are generally made by chemically modifying substances that are
available from natural source to improve its potency, efficacy and/or reduce side
effects. Sometimes, semi-synthetic processes are used to prepare drugs when
the natural sources may yield impure compounds or when the synthesis of drugs
(complex molecules) may be difficult, expensive, and commercially unviable. When
the nucleus of drug obtained from natural source is retained but the chemical
structure is altered, we call it semi - synthetic. E.g. Apomorphine, Diacetyl morphine,
Ethinyl Estradiol, Homatropine, Ampicillin and Methyl testosterone7. Microbiological sourcesSeveral life-saving drugs have been historically derived from microorganisms.
Examples include penicillin produced by Penicillium chrysogenum,
streptomycin from Streptomyces griseus, chloramphenicol from Streptomyces
venezuelae, neomycin from Streptomyces fradiae, bacitracin from Bacillus
subtilis etc.
PenicilliumNotatum is a fungus which gives penicillin, Actinobacteria gives
Streptomycin, and Aminoglycosides such as gentamicin and tobramycin are
obtained from streptomycis and micromonosporas.8. Recombinant DNA technology/ Biosynthetic sources (genetically
engineered drugs)This is relatively a new field which is being developed by mixing discoveries fromDNA by enzyme restriction endonucleases. The desired gene is coupled to rapidly
molecular biology, recombinant DNA technology, DNA alteration, gene splicing,
immunology, and immune pharmacology. Drugs developed using living organisms
with the help of biotechnology or genetic engineering are known as biologics,
biopharmaceuticals, recombinant DNA expressed products, bioengineered, or
genetically engineered drugs Examples include recombinant Hepatitis B vaccine,
recombinant insulin and others. Recombinant DNA technology involves cleavage of
replicating DNA (viral, bacterial or plasmid). The new genetic combination is inserted
into the bacterial cultures which allow production of vast amount of genetic material.
Advantages: Huge amounts of drugs can be produced, Drug can be obtained in
pure form, and It is less antigenic (induce immune system). Disadvantages: Well-
equipped lab is required, highly trained staff is required and it is a complex and
complicated technique.Self- assessment 1.11. The use of the drug started when?2. What are the sources of drug?
3. Who first isolated morphine from opium in 1805?
4. Who is considered as the father of American pharmacology?1.2 Definition of key termsLearning activity 1.2As the new student admitted in Associate nurse program in senior 4, read the
book of pharmacology and define the following common key terms used in
pharmacology:
1. Pharmacology
2. Clinical pharmacology
3. Drugs
4. Adverse drug reaction and
5. Therapeutic effectCONTENT SUMMARYPharmacology: The word pharmacology is derived from two Greek words,
“pharmakon”, which means medicine or drug, and logos, which means study. It
is the study of medicines. It includes the study of how drugs are administered and
how the body responds (Adams et all 2014).It can be also defined as the study of drugs and their interactions with living systems.Clinical pharmacology: is defined as the study of drugs in humans.Drugs: chemicals that are introduced into the body to bring about some sort ofchange.Adverse drug reaction: Any unexpected, unintended, undesired, or excessive
response to a medication given at therapeutic dosages (Linder et al 2014).Drug actions: The processes involved in the interaction between a drug and body
cells (e.g., the action of a drug on a receptor protein); also called mechanism of
action.Drug classification: A method of grouping drugs; may be based on structure or
therapeutic use.Drug effects: The physiologic reactions of the body to a drug. They can be
therapeutic or toxic and describe how the body is affected as a whole by the drug.
The terms onset, peak, and duration are used to describe drug effects (most often
referring to therapeutic effects).Pharmacognosy The study of drugs that are obtained from natural plant and animal
sources.Therapeutic effect: The desired or intended effect of a particular medication.
Therapeutic index: The ratio between the toxic and therapeutic concentrations of
a drug.Tolerance: Reduced response to a drug after prolonged use.Toxic: The quality of being poisonous (i.e., injurious to health or dangerous to life).Toxicity: The condition of producing adverse bodily effects due to poisonous
qualities.Food and Drug Administration (FDA): federal agency responsible for the
regulation and enforcement of drug evaluation and distribution policiesSelf- assessment 1.2Define the following terms:1. Pharmacognosy
2. Therapeutic index
3. Tolerance1.3 Chemical drug nameLearning activity 1.31. Read the book of pharmacology and explain chemical drug name (using
library textbook)CONTENT SUMMARYDrugs are chemicals that are introduced into the body to bring about some sort of
change. The drugs have several names, which may cause confusion. Each drug
has three names: a chemical name, a generic name, and a brand name. The
health care professionals have to study pharmacology which is the study of drugs
and their interactions with living systems to know the exact medication to be used
and to control the complication associated.The chemical names are the scientific names, based on the molecular structure of
the drug. There are various systems of chemical nomenclature and thus various
chemical names for any one substance. The most important is the International
Union of Pure and applied Chemistry (IUPAC) name. A drug has only one
chemical name. Chemical names are typically very long and too complex to
be commonly used in referring to a drug in speech or in prose documents. For
example, “1-(isopropylamino)-3-(1-naphthyloxy) propan-2-ol” is a chemical name
for propranolol. Sometimes, a company that is developing a drug might give the
drug a company code, which is used to identify the drug while it is in development.
This chemical name is sometimes helpful in predicting a drug’s physical and
chemical properties. Examples of chemical names of common drugs include
lithium carbonate, calcium gluconate, and sodium chloride.Self- assessment 1.31. A drug can have different name. Which one among the following drug
name is chemical name?
a. N-acetyl-p-aminophenl
b. paracetamol
c. Tylenol2. A drug has how many chemical name?3. Give 3 examples of easy chemical names to remember of common drugs1.4 Generic drug nameLearning activity 1.4Your neighbour sent her child to the pharmacy to buy the Paracetamol tablets.
The pharmacist gives the child the firm coated tablet labelled as PANADOL®.
The neighbour becomes confused and returns to the pharmacy for clarification
before taking the drug. The pharmacist tells the neighbour that, it is the same drug.
One is generic name (Paracetamol) and the other is brand name (Panadol®)1. Give the difference between generic name and brand name.CONTENT SUMMARY
The generic name is simpler name, less complicated and easier to remember
than chemical names. It may be used in any country and by any manufacturer. The
first letter of the generic name is not capitalized. Students are strongly encouraged
to learn and refer to drugs by their generic names because formularies (i.e., lists of
medicines available through a pharmacy) are maintained by generic names. When
a therapeutically equivalent drug becomes available in generic form, the generic
medicine is routinely substituted for the brand-name medicine. Generic names are
provided by the United States Adopted Names Council, which is an organization
sponsored by the United States Pharmacopeial Convention, the American Medical
Association, and the American Pharmacists Association. The official name, which
is virtually always the generic name in the United States, is the name under which
the drug is listed by the US Food and Drug Administration (FDA). The FDA is
empowered by federal law to generically name the drugs for human use in the
United States.Food and Drug Administration (FDA) is federal agency responsible for the
regulation and enforcement of drug evaluation and distribution policies. Because
there is only one generic name for each drug, health care providers often
use this name and they must memorize it. Generic drugs are less expensive
than brand-name drugs, but they may differ in bioavailability. Bioavailability
is defined by the Federal Food, Drug and Cosmetic Act as the rate and extent to
which the active ingredient is absorbed from a drug product and becomes available
at the site of drug action to produce its effect. Bioavailability may be affected by
many factors, including inert ingredients and tablet compression. Anything
that affects the absorption of a drug or its travel to the target cells can certainly
affect drug action. Measuring how long a drug takes to exert its effect (onset time)
gives pharmacologists a crude measure of bioavailability. If the trade and generic
products have the same rate of absorption and have the same onset of therapeutic
action, they are said to be bioequivalent.Self- assessment 1.41. A patient/client tells to the nurse that is taking aspirin. Which type of drug
name is this?2. A drug can have different name. Which one among the following drug
name is generic name:
a. (RS)-2-(4-(2methylpropyl)phenyl) propanoic acid
b. ibuprofen
c. Motrin3. _____ means that the amount of active ingredient that reaches the
patient’s bloodstream for a generic drug must be equivalent to that of the
branded drug.
a. Bioequivalence
b. Route of administration
c. Monitoring of adverse events
d. Biohazard labels4. What does the term “bioavailability” mean?
a. Plasma protein binding degree of substance
b. Permeability through the brain-blood barrier
c. The rate and extent to which the active ingredient is absorbed
d. Amount of a substance in urine relative to the initial doze1.5 Trade drug nameLearning activity 1.5Read the book of pharmacology and explain trade drug name (using library
textbook)A drugs trade name, sometimes called the proprietary, product, or brand name,
is assigned by the pharmaceutical company marketing the drug and it is followed by
the symbol ®. This symbol indicates that the name is registered and that the use of
the name is restricted to the owner of the drug, which is usually the manufacturer.
The trade name is intentionally selected to be short and easy to remember so that
patients will remember it (and ask for it by name).Drugs with more than one active generic ingredient are called combination drugs.
Acetaminophen and aspirin are examples of agents that appear in many combinationdrugs with dozens of different trade names. To avoid this confusion, generic
names should be used when naming the active ingredients in a combination
drug. When referring to a drug, it is conventional to write the generic name in lower
case first, followed by the trade name in parentheses with the first letter capitalized.
Examples include alprazolam (Xanax) and acetaminophen (Tylenol). (Cyton et al
2017).The difference between trade name and trademark name is that a trade name
refers to the company’s official name, while a trademark provides a company’s
brand with legal protection.The key to comparing brand-name drugs and their generic equivalents lies in
measuring the bioavailability of the two agents. Bioavailability is defined by the
Federal Food, Drug and Cosmetic Act as the rate and extent to which the active
ingredient is absorbed from a drug product and becomes available at the site of
drug action to produce its effect. Bioavailability may be affected by many factors,
including inert ingredients and tablet compression. Anything that affects the
absorption of a drug or its travel to the target cells can certainly affect drug action.
Measuring how long a drug takes to exert its effect (onset time) gives pharmacologists
a crude measure of bioavailability. If the trade and generic products have the same
rate of absorption and have the same onset of therapeutic action, they are said to
be bioequivalent.The importance of bioavailability differences between a trade name drug and its
generic equivalent depend on the specific circumstances of pharmacotherapy. For
example, if a patient is in circulatory shock and the generic equivalent drug takes
5 minutes longer to produce its effect that may indeed be significant. However, if a
generic medication for arthritis pain relief takes 45minutes to act, compared to the
brand-name drug that takes 40 minutes, it probably does not matter which drug is
used, and the inexpensive product should be prescribed to provide cost savings to
the consumer.As a general rule, bioavailability is of most concern when using critical care
drugs and those with a narrow safety margin. In these cases, the patient should
continue taking the brand name drug and not switch to a generic equivalent, unless
approved by the health care provider. For most other drugs, the generic equivalent
may be safely substituted for the trade name drug.In the age of Internet pharmacies, the issue of exclusive marketing rights has
drastically changed. In some cases, they even sell the drug to consumers without
a prescription. Other countries do not have the same quality control standards as
the United States, and the patient may be purchasing a useless or even harmful
product. Furthermore, although Internet sites may appear to be based in the United
States, they may instead be obtaining their medications from unreliable sources.Nurses must strongly urge their patients not to purchase drugs from overseas
pharmacies because there is no assurance that the drugs are safe or effective.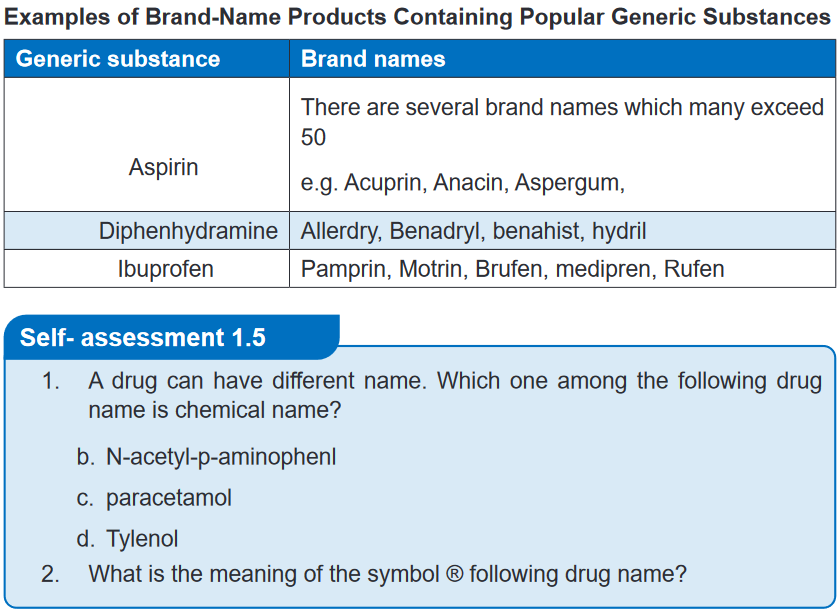 1.6. Label of drugs containerLearning activity 1.6
1.6. Label of drugs containerLearning activity 1.6 The Food and Drug Administration have specific information that identifies a specific
The Food and Drug Administration have specific information that identifies a specific
drug. It is important to obtain a thorough and accurate information from the drug
containers regarding their labelling, as they can often provide valuable information.The Drug label is a standardized label that appears on all over-the-counter
(OTC) medicines approved by the Food and Drug Administration, it have specific
information that identifies a specific drug. It is designed to tell the purpose of the
medicine, who should take the medicine and how to take it safely. For example, a
drug label identifies the brand and generic names for the drug, the drug dosage,
the expiration date, and special drug warnings. Some labels also indicate the route
and dose for administration. It’s very important to read all the information on the
label and current and approved references every time someone want to administer
a medicine because labels change regularly. Nurses need to become familiar with
each aspect of the label.A lack of information on drug labelling can result in serious mistakes in the
preparation of drugs which can place patients at risk. In all drug packaging of
the chosen drug should contain; what drug is to be used, how the drug is taken,
when the drug is to be administered, the importance of taking the drug (patient
compliance) and information about what happens if it is not taken as prescribed
(patient noncompliance), how long the drug is to be used, what adverse effects can
be expected and the alternatives available. The term compliance is the extent to
which patients follow instruction.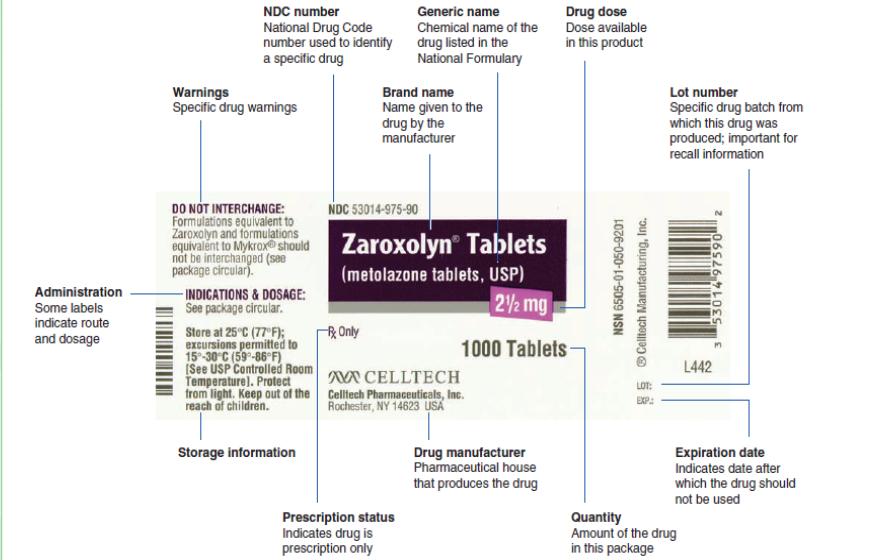 Numerous drug labels are used in the drug calculation problems to familiarize the
Numerous drug labels are used in the drug calculation problems to familiarize the
nurse with important information on a drug label. This information is then used in
correctly calculating the drug dose.Self- assessment 1.61. Enumerate the necessary information which must be on the drug
containers?2. What is drug label?3. During the drug administration the nurse found a Drug container on which
the label information is not clear?1.7. Solid drug dosage formsLearning activity 1.7 1. What do you observe on this image?
1. What do you observe on this image?
2. Are there any other solid forms of medication which are not on this image?
3. Why is it important for nurses to know different type of solid drug dosage
form?CONTENT SUMMARYSolid dosage forms include tablets, capsules, Caplets, Lozenges/ troches,
Powders and granules. The Tablets are available in variety of sizes, shapes,
colors, and thicknesses, usually obtained by single or multiple compressions of
powders or granules.Most tablets and caplets are designed to be swallowed whole and dissolve in the
gastrointestinal tract, but some are also made to be administered sublingually,
buccally, or vaginally.Tablets are normally right circular solid cylinders, the end surfaces of which are flat
or convex and the edges of which may be beveled. They may have lines or break-
marks (scoring), symbols or other markings.4. Uncoated tablets: compressed tablet or core tablet formed by compression
and contain no special coating. They are made from active ingredient in
combination with excipients such as binders, diluents, etc. Example: Analgin,
Paracetamol, Bactrim, etc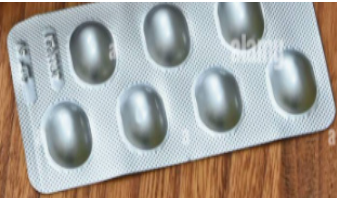 5. Sugar-coated tablets: are smooth, round or oval compressed tablets
5. Sugar-coated tablets: are smooth, round or oval compressed tablets
containing sugar coatings. Sugar coating provides both protection and sweet
taste but the coating operations take a long time. Example: Neocodion,
Paderyl, Aphatyl, Spasfon etc. 6. Film-coated tablets: are compressed tablets which are covered with a thin
6. Film-coated tablets: are compressed tablets which are covered with a thin
layer or a film of polymeric substances to protect their contents from moisture
or to mask the taste of the ingredients. Example: Ibuprofen 7. Modified release tablet: Modified-release tablets are coated, uncoated
7. Modified release tablet: Modified-release tablets are coated, uncoated
or matrix tablets containing excipients or prepared by procedures which,
separately or together, are designed to modify the rate, the place or the time
of release of the active ingredient(s) in the gastrointestinal tract. Sustained-
release tablets (Extended/Controlled/Prolonged-release): Sustained-release
tablets are designed to slow the rate of release of the active ingredient(s) in
the gastrointestinal tract.8. Example: Nifedipine. Delayed-release tablets (Entered-coated/Gastro-
resistant tablets): are coated with substances that resist solution in gastric
fluid but disintegrate in the alkaline contents of the intestine. Enteric coating
is used for medicines with a gastric irritant action, for medicines which areunstable in the acid medium of the gastric contents or if the medicine should
act on the intestine. Example: Aspirin 81mg.9. Effervescent tablets: Effervescent tablets are uncoated tablets generally
containing acid substances and carbonates or hydrogen carbonates that
react rapidly in the presence of water to release carbon dioxide. They
are intended to be dissolved or dispersed in water before administration.
Example: Efferalgan Vitamin C, Berroca Suppradine, etc 10. Chewable tablets: Chewable tablets are usually uncoated. They are intended
10. Chewable tablets: Chewable tablets are usually uncoated. They are intended
to be chewed before being swallowed; however, where indicated on the
label, they may be swallowed whole instead. They should be hard and large
which difficult to swallow. Example : Maalox, Amoxicillin chewable zentel,
etc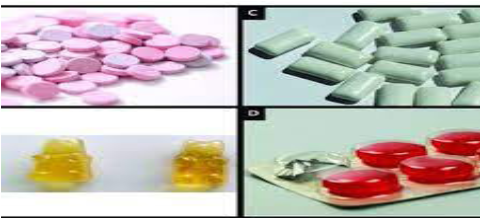 11. Lozenge tablets (Troche): Tablets containing palatable flavoring, indicated
11. Lozenge tablets (Troche): Tablets containing palatable flavoring, indicated
for a local (often soothing) effect on the throat and mouth. They are placed
in the mouth where they slowly dissolve, liberating the active ingredient. The
drug involved can be antiseptic, local anesthetic, antibiotic, or antitussive.
Example: Lysopaine, Horf, Strepsil, Wood, Zecuf, etc. Patient is advised not
to swallow a lozenge; it should be allowed to slowly dissolve in the mouth.
Patient is also advised not to drink liquids for approximately 15 minutes after
administration, to prevent washing of the lozenge contents from throat or
mouth. 12. Sublingual tablets: Medicine is placed under the tongue and allows dissolving.
12. Sublingual tablets: Medicine is placed under the tongue and allows dissolving.
It is absorbed into the circulation and provides the systemic effects. This
medication form is suitable for the active ingredients which is destroyed or
unstable in the gastrointestinal fluids. Example: NitroglycerinCapsules are solid dosage forms in which the drug substance is enclosed in either
a hard or soft soluble container of suitable form of gelatin. They are intended to
mask the smell and taste of the drug substances. Capsules are tasteless, easily
administered and some patients prefer them to the tablets. They are of various
shapes and sizes and contain a single dose of one or more active ingredients.
Capsules may be Hard Gelatin Capsules, Soft Gelatin Capsules and Modified-
Release Capsules:1. Hard Gelatin Capsules: Hard capsules have shells consisting of two
prefabricated cylindrical sections that fit together. One end of each section
is shorter, larger rounded, and closed (cap) and the other is open, longer
and smaller (body). The contents of hard capsules are usually in solid form
(powder or granules). Example: Amoxicillin, Ampicillin, Cephalexin, etc.2. Gelatin Capsules: Soft capsules have thicker shells than hard capsules and
antimicrobial preservatives are usually added. The shells are of one piece
and various shapes. They may be round, oval and oblong. The contents of
soft capsules are usually solutions or suspensions of the active ingredient(s)
in non-aqueous liquids. Example: Vitamin E, Vitamin A, Eugica, etc3. Modified-Release Capsules: Modified-release capsules are hard or soft
capsules in which the contents or the shell or both contain excipients or
are prepared by special procedures such as micro-encapsulation which,
separately or together, are designed to modify the rate, place or time of
release of the active ingredient(s) in the gastrointestinal tract. Sustained-
release capsules are designed to slow the rate of release of the active
ingredient(s) in the gastrointestinal tract. Example: Cardene SR (nicardipine).
Delayed-release capsules are hard or soft capsules prepared in such
a manner that either the shell or the contents resist the action of gastric
fluid but release the active ingredient(s) in the presence of intestinal fluid.
Examplse: Casprin, Esomeprazole4. The caplets/ Pills are small, round dosage forms for oral administration
which are prepared by the pharmacist. They are rarely prescribed today.
The powdered ingredients are mixed together with binding agents. The pill
mass is rolled into spheres and coated with talc, gelatin, or sugar.Example: Oral contraceptive pills.5. Oral Powder: oral powders are preparations consisting of solid, loose, dry
particles of varying degrees of fineness. They contain one or more active
ingredients, with or without excipients and, if necessary, authorized colouringmatter and flavouring substances. They are generally administered in or
with water or another suitable liquid. They may also be swallowed directly.
They are presented as single-dose or multidose preparations. Each dose of
a single-dose powder is enclosed in an individual container, for example a
packet, a sachet or a vial. Multidose oral powders require the provision of a
measuring device capable of delivering the quantity prescribed.Example: Clamoxyl 250mg, Dolipran, powder, Smecta, etc.6. Granules are dosage forms related to powders. They are particularly suitable
for the preparation of solutions or mixtures of medicines. Example: Montiget,
Biorrhee, etc.Self- assessment 1.71. The associate nurse students are reviewing principles of pharmacology,
and are reading about different forms of drugs. Enumerate 6 solid drug
dosage forms which can be used orally?2. Some tablets to treat a headache must first be dissolved in water before
swallowing. Which one of the following best describes this type of tablet?
a. Modified release
b. Oral disintegrating
c. Effervescent
d. Buccal3. Capsules in which powders are enclosed are made up of …..
a. Gelatine
b. Rice flour
c. Fructose
d. Dextrose
1.8. Semisolid drug dosage forms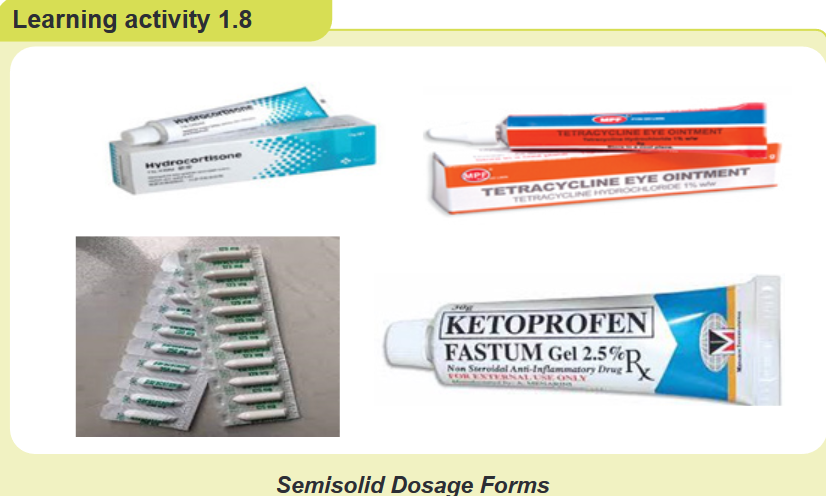 1. What do you observe on this image?
1. What do you observe on this image?
2. Are there any other semisolid forms of medication which is not on this
image?
3. Why is it important for nurses to know different type of semisolid drug
dosage form?CONTENT SUMMARY
Semisolid dosage forms are normally presented in the form of creams, gels,
ointments, pastes, suppository or patch. They contain one or more active ingredients
dissolved or uniformly dispersed in a suitable base and any suitable excipients
such as emulsifiers, viscosity-increasing agents, antimicrobial agents, antioxidants,
or stabilizing agents. The choice of a base for semi-solid dosage forms depends on
many factors: the therapeutic effect desired the nature of the active ingredient to be
incorporated, the availability of the active ingredient at the site of action, the shelf-
life of the finished product, and the environmental conditions in which the product
is intended to be administered.It should be smooth, inert, odorless, physically and chemically stable, and
compatible with both the skin and the active ingredient(s) to be incorporated. It
should normally be of such a consistency that it spreads and softens easily when
stress is applied. It may be necessary for a topical semi-solid dosage form to be
sterile, for example, when it is intended for use on large open wounds or severely
injured skin.Creams are homogenous, semisolid preparation that is usually white and no
greasy and has a water base. Creams are intended for application to the skin
or certain mucous membranes for therapeutic or protective purposes. The term
“cream” is most frequently used to describe soft, cosmetically acceptable types of
preparations.Example: Hydrocortisone cream, Ketoconazole cream, etc Ointments are homogeneous, semi-solid and greasy preparations intended for
Ointments are homogeneous, semi-solid and greasy preparations intended for
external application to the skin or mucous membranes for therapeutic or protective
purposes.Example: Tetracycline ointment. Gels are usually homogeneous, clear, semi-solid, jelly- like preparations that may
Gels are usually homogeneous, clear, semi-solid, jelly- like preparations that may
be used for topical medication. Gels are applied to the skin or certain mucous
membranes for therapeutic, or protective purposes.Example: Erythrogel, fastum gel, etc Pastes are homogeneous, semi-solid preparations containing high concentrations
Pastes are homogeneous, semi-solid preparations containing high concentrations
of insoluble powdered substances (usually not less than 20%) dispersed in a
suitable base. The pastes are usually less greasy, more absorptive, and stiffer inconsistency than ointments because of the large quantity of powdered ingredients
present. Pastes adhere reasonably well to the skin and they are suited for application
on and around moist lesions.Example: Orrepaste, Anagelsic and anti-inflammatory containing dental paste, etc Patched/Plasters are substances intended for external application made of such
Patched/Plasters are substances intended for external application made of such
materials and of such consistency as to adhere to the skin. Inner surface of the
patch contacts skin and allows transdermal absorption of lipid-soluble medicines.
The total amount of medicine on the patch is very large, but typically only a small
fraction is absorbed. Patch are convenient because they can be applied easily and
minimize stomach upset. They can also improve compliance because there is no
need for more frequent dosing like oral dosage forms.Example: Dermal patches Suppository are semisolid dosage forms to be inserted into body cavity-rectum or
Suppository are semisolid dosage forms to be inserted into body cavity-rectum or
vagina, where medication is melt at the body temperature which provides local or
systemic effects.Example: paracetamol suppository, Flagyl suppository, etc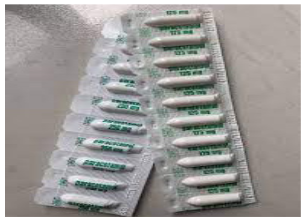 Advantage of semisolid dosage form are: It is used externally, the probability of side
Advantage of semisolid dosage form are: It is used externally, the probability of side
effects can be reduced, first-pass gut and hepatic metabolism is avoided, local action
and site-specific action of the drug on the affected area, convenient for unconscious
patients or patients to have difficulty in oral administration, suitable dosage form for
bitter drugs and more stable than a liquid dosage form. The disadvantage of using
semisolid drug forms are: The accuracy can’t be measured, for the semisolid dosage
form, may cause staining, they are bulky to handle, application with a finger may
cause contamination, physico-chemical is less stable than a solid dosage form and
may cause irritation or allergy to some patients. The ideal properties of semisolid
dosage forms are smooth texture, elegant in appearance, non-dehydrating, non-
gritty and non-greasy and non-staining and Non-hygroscopic.Self- assessment 1.81. Enumerate the semisolid dosage forms.
2. Which of the following is not a semisolid dosage form
a. Paste
b. Cream
c. Ointments
d. Suspension
3. A semi-solid preparations containing high concentrations of insoluble
powdered substances (usually not less than 20%) dispersed in a suitable
base is known as:
a. Paste
b. Suppository
c. Ointments
d. Gels CONTENT SUMMARY
CONTENT SUMMARY
Liquid dosage forms are prepared by dissolving the active ingredient(s) in an
aqueous or nonaqueous solvent, by suspending the drug in appropriate medium
or by incorporating the drug substance, into one or two phases of an oil and water
system. These forms can be formulated for different routes of administration: oral
use, introduction into body cavities, or applied externally. Liquid drugs may also be
administered systemically by mouth or by injection throughout the body.The oral liquid forms can be readily administered to children or people unable to
swallow tablets or capsules.Syrup is a medicine dosage form that consists of a high concentration of a sugar
in water. Flavors may be added to mask unpleasant taste of certain medication.
Cherry, grape, strawberry syrup drug preparations are common for children.
Example: Sara syrup, Ibuprofen syrup, Dalfagan syrup, etc.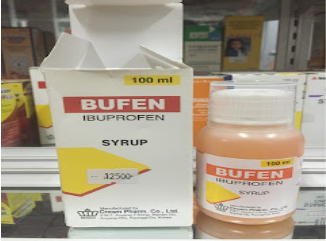 Suspension is liquid form of medication that must be shaken well before
Suspension is liquid form of medication that must be shaken well before
administration because the medicine particles settle at the bottom of the bottle. The
medicine is not evenly dissolved in the liquid (hydrophobic agents). Example: Cotrim
suspension, Diaryl suspension, Amoxicillin suspension, Cefixim suspension, etc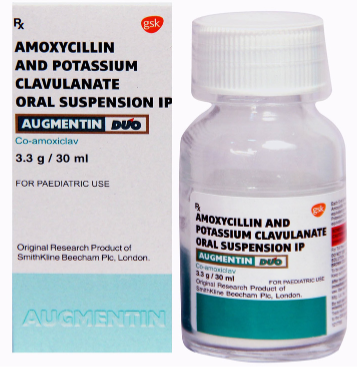 Elixir is liquid medicine form for oral use that contain primarily water, alcohol and
Elixir is liquid medicine form for oral use that contain primarily water, alcohol and
sugar. Their alcohol content makes elixir convenient liquid dosage form for many
drugs that are only slightly soluble in water. Example: Hosolvan elixir, Terpin hydrate
elixir, etc. Emulsion is a pharmaceutical preparation in which two agents of oil and water that
Emulsion is a pharmaceutical preparation in which two agents of oil and water that
cannot ordinarily be combined are mixed. These forms can be administered orally,
topically, or parenterally (intramuscularly). In order to prepare suitable emulsions
and to have them remain stable for a suitable period of time, a number of emulsifying
agents are used in their preparation. Example: Propofol (Diprivan), Metronidazole
topical emulsion, etc. Tincture is alcoholic or water- alcohol solution of medicines. It differs from elixir
Tincture is alcoholic or water- alcohol solution of medicines. It differs from elixir
in that it is not sweeten. Tincture can be used orally or externally. Example: Iodine
tincture Eye, Ear and Nose Drops are medicines in sterile water (purified water-deionized,
Eye, Ear and Nose Drops are medicines in sterile water (purified water-deionized,
demineralized water) to be applied by drops.Example: Ciprofloxacin eye/ear drop, New V-rotho, Tear Natural II, Pyinchin, etc.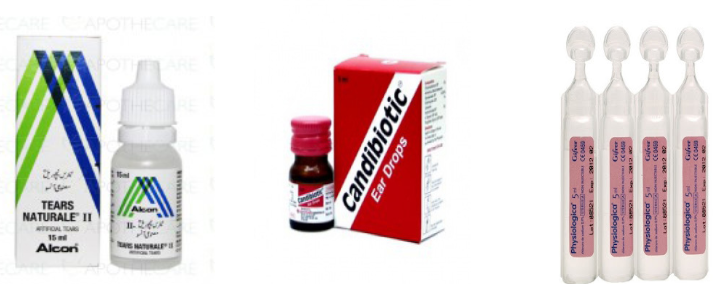 Mouth washes solution is aqueous solutions which are most often used for their
Mouth washes solution is aqueous solutions which are most often used for their
deodorant, refreshing or antiseptic effect.Example: Eludril, Septil, etc Enema is a fluid injected into the lower bowel by way of the rectum and most
Enema is a fluid injected into the lower bowel by way of the rectum and most
frequent used as a cleansing enema which is given to relieve constipation or for
bowel cleansing before a medical examination or procedure.Example: Pata enema, etc Douche solution is sterile solution, often a cleansing or antiseptic agent for part of
Douche solution is sterile solution, often a cleansing or antiseptic agent for part of
the body or body cavity.Example: Povidone iodine, H2O2 Liniment is the preparation for external use that is rubbed on the skin as a
Liniment is the preparation for external use that is rubbed on the skin as a
counterirritant. As such, the liniment creates a different sensation (e.g. tingling or
burning) to mask pain in the skin, muscle or joint.Example: Camphor liniment Medications for injection: solution have a sterile water base and are thus referred
Medications for injection: solution have a sterile water base and are thus referred
to as aqueous solution. Some solutions have an oil base, which tends to cause a
more prolonged absorption time. The oily nature of these solutions makes them
thick, thus they are referred to viscous solution.Example: Becozyme injection, Glucose 50% injection, Lactate ringer, NaCl 0.9%,
etc Powder are dry particle of medicines. The powder itself cannot be injected. It must
Powder are dry particle of medicines. The powder itself cannot be injected. It must
be mixed with a sterile diluting solution-solvent (sterile water or saline solution) to
render an injectable solution. This is termed reconstitution of medicine. Medicines
are supplied undiluted in powder form because of the short period of time they
remain stable after dilution.Example: Ampicillin, Ceftriaxone, etcThese products are packaged in ampoules, vials, bottles, plastic bags, and prefilled
disposable syringes. Self- assessment 1.91. Give the difference between suspension and emulsion2. Which of the following formulations would not be applicable to ocular
Self- assessment 1.91. Give the difference between suspension and emulsion2. Which of the following formulations would not be applicable to ocular
administration?
A. Solution
B. Liniment
C. Suspension
D. Ointment3. The component present in solution in small quantity is known as…..
A. Solvent
B. Solution
C. Solute
D. Liquid4. The component present in solution in large quantity is known as.
A. Solvent
B. Solution
C. Solute
D. Liquid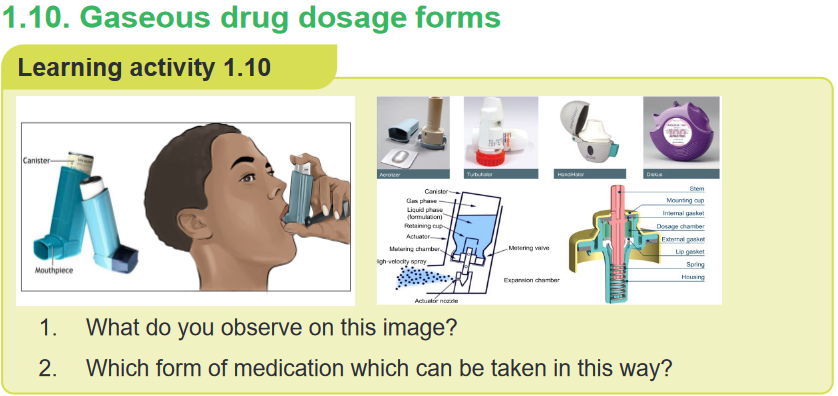 In gaseous dosage forms, the active pharmaceutical ingredients (API) are given
In gaseous dosage forms, the active pharmaceutical ingredients (API) are given
in the form of gas, are packed in a special container which gets released upon
applying pressure. It is used in the nose and mouth for local application or on the
skin. This allows medicines to be delivered to and absorbed in the lungs, which
provides the ability for targeted medical treatment to this specific region of the body,
as well as a reduction in the side effects of oral medications.E.g. Inhalers, aerosols, vaporizers, sprays, and nebulizers or atomizersAerosols are suspension of fine solid or liquid particles with gas used to apply drug
to respiratory tract having atomizer with in device. Inhalations are internal liquid
preparations containing medicaments dissolved in suitable solvent or if insoluble
suspended in the propellantSprays are Gaseous preparations of drugs containing alcohol applied to mucous
membrane of nose or throat with atomizer or nebulizer.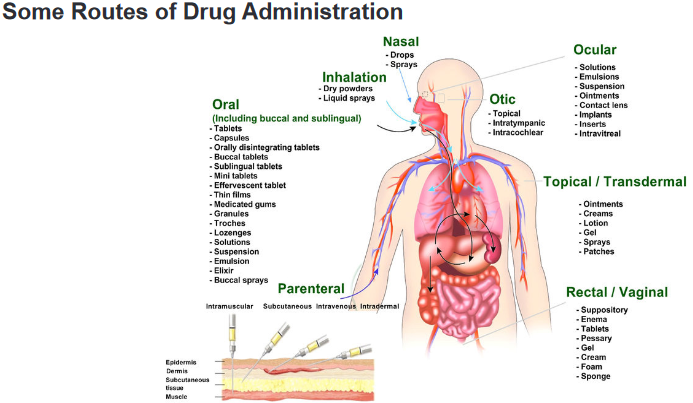 Self- assessment 1.101. Enumerate the routes of administration of Gaseous dosage forms
Self- assessment 1.101. Enumerate the routes of administration of Gaseous dosage forms
2. What are the difference between aerosols and sprays?1.11 Dose and drug regimenLearning activity 1.10 1. What do you observe on this image?
1. What do you observe on this image?
2. Explain the importance of taking the medication as prescribed?
3. What is the effective dose?
4. Based on this image what do thing you will learn in this unit?
Learning activity 1.10The patient diagnosed with disease has to take the medications as prescribed by
the authorised health professional. To avoid and to achieve desirable therapeutic
effect, the patient has to take the correct dose. A dose refers to a specified amount
of medication taken at one time while the dosage is the prescribed administration
of a specific amount, number, and frequency of doses over a specific period of time.
A dosage guides a drug regimen.A drug regimen is a prescribed systematic form of treatment for a course of
drug(s). Regimen is a treatment plan that specifies the dosage, the schedule, and
the duration of treatment. Dose regimen includes the loading dose, maintenance
dose, dose frequency, dose duration, and dose adjustments for special populations
and for coadministration with other drugs.The drugs dose can be given as single dose, continuous administration and
irregular or several doses administration. The Single dose: After an intravenous
injection, the drug enters the bloodstream directly and the concentration rises to its
peak level almost immediately. Elimination and distribution will start immediately.With intramuscular injection, the drug is absorbed over a longer period, and following
oral administration, absorption takes even longer. The effect of a drug is usually
fastest if the route of administration that leads most rapidly to a high concentration
in the target organ is used.Continuous administration – intravenous infusion if a drug is administered by
a continuous intravenous infusion, the absorption phase will last as long as the
infusion continues.Irregular administration – several doses per day If a drug is administered in
‘portions’, or by several doses per day, the absorption and subsequent concentration
of the drug in the blood will vary between each dose. Initially, the concentration
increases for each new dose, if the time interval between the doses is so short that
the drug is not totally eliminated before the next dose is taken.This increase in concentration gradually diminishes, and steady state is eventually
achieved, as the rate of elimination of drug increases with increased concentration
of the drug. Once steady state is achieved, the concentration of the drug will only
vary between doses. The concentration rises immediately after intake, reaches a
peak level, and drops gradually until the next dose is taken.Even though many people receive the same dose of a drug, not all of them will
achieve the same effect. Some may have effect with a low dose, while others require
a higher dose. Likewise, some notice adverse effects at lower doses than others.The Effective dose is the dose that produces the desired effect. Based on the
amount the client received the dose can be effective, toxic and lethal dose. The
toxic dose is the dose that produces a toxic effect. The lethal dose is the dose
that results in death. This is an experimental term that can only be determined
in animal experiments and estimated in humans taking high doses in attempting
suicide.Drug dosage errors can occur at any time from when the drug is prescribed to its
administration and mistakes can place patients at risk; at worst, they can be fatal.
The cause of drug dosage errors can be attributed to both the health professional
and the patient. When using drugs with potent effects, it is even more important to
have a raised awareness, to avoid potential dosage errors. The same applies when
administering drugs to small children, the elderly and unconscious patients.A loading dose is a higher amount of drug, often given only once or twice, that is
administered to “prime” the blood-stream with a level sufficient to quickly induce
a therapeutic response. Before plasma levels drop back toward zero intermittent
maintenance doses are given to keep the plasma drug concentration in the
therapeutic range. Although blood levels of the drug fluctuate with this approach,
the equilibrium state can be reached almost as rapidly as with a continuous infusion.When immediate drug response is desired, a large initial dose, known as the loading
dose, of drug is given to achieve a rapid minimum effective concentration in the
plasma. After a large initial dose, a prescribed dosage per day is ordered.Loading doses are particularly important for drugs with prolonged half-lives and
for situations in which it is critical to raise drug plasma levels quickly, as might be
the case when administering an antibiotic for a severe infection. It took almost five
doses (48hours) to reach a therapeutic level using a routine dosing schedule. With
a loading dose, a therapeutic level can be reached within 12 hours.Maintenance doses are the dose taken to maintain the plasma concentration.
During the long-term use of some drugs, it is customary to prescribe fixed doses
with virtually identical long intervals between doses. With a dosage of 1 ×1, there
will be 24 h between each dose. With a dosage of 1 ×3, there will be 8 h between
each dose. With dosages that are more frequent than twice a day, the dosage
intervals will, in practice, often vary during the course of the day. Maintenance dose
can be also administered after loading dose to maintain the plasma concentration
of the drug.Self- assessment 1.111. Give the difference between the loading dose and maintenance dose
2. What are the difference between dose and dosage?
3. A client was diagnosed with malaria and is taking quinine by oral route.
The medical prescription indicate that the patient will take 10mg per Kg
per day in 3 times (every 8hours). Explain why the patient have to take
the medication every 8 hours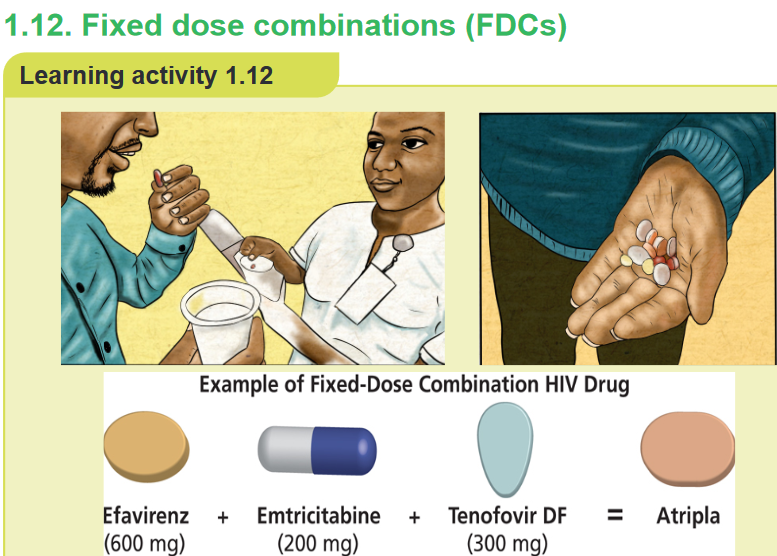 1. What do you observe on this image?
1. What do you observe on this image?
2. Atripla is combination of which drug?CONTENT SUMMARY
Good adherence to medication is one of the cornerstones of successful management
of chronic diseases. Unfortunately, such adherence is often difficult to achieve and
estimates suggest that only 50% of all chronic disease patients are able to adhere
to treatment. Fixed dose combinations (FDCs) are defined as a combination of two
or more active ingredients within a single form of pharmaceutical administration.They have been shown to appreciably reduce the risk of medication non adherence,
which is particularly important in patients with chronic diseases. An example of
a fixed-dose combination (FDC) HIV drug is Atripla (a combination of efavirenz,
emtricitabine, and tenofovir disoproxil fumarate) and Bactrim (sulfamethoxazole +
trimethoprim). By reducing the number of pills a person must take each day, fixed-
dose combination drugs can help improve adherence to treatment regimen.It is widely accepted that most drugs should be formulated as single compounds.
Fixed ratio combination products are acceptable only when the dosage of each
ingredient meets the requirement of a defined population group and when the
combination has a proven advantage over single compounds administered
separately in therapeutic effect, safety or complianceThe rationality of FDCs should be based on certain aspects such as: The drugs in
the combination should act by different mechanisms, the pharmacokinetics must
not be widely different and the combination should not have supra-additive toxicity
of the ingredients. 1.13 Directly observed therapy (DOT)Learning activity 1.13
1.13 Directly observed therapy (DOT)Learning activity 1.13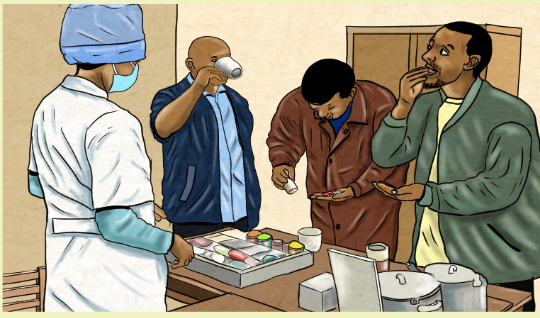 1. What do you observe on this image?
1. What do you observe on this image?
2. Explain why it is necessary to take drug while the nurse is observingCONTENT SUMMARY
Directly observed therapy (DOT) is used to ensure that the person receives
and takes all medications as prescribed and to monitor response to treatment.
DOT is widely used to manage tuberculosis (TB) disease. In HIV treatment, DOT is
sometimes called directly administered antiretroviral therapy (DAART).The World Health Organization (WHO) and the Centers for Disease Control and
Prevention (CDC) recommend directly observed therapy (DOT) for TB treatment
to monitor and provide treatment support for affected people whenever feasible.
When implemented properly, DOT fosters high levels of treatment adherence and
early detection of adherence problems, adverse drug reactions, and worsening TB
symptoms.Even if a proposed drug treatment is the optimal choice for a disease, it will not be
effective without patient compliance (the extent to which patients follow instructions).There are causes and many possible reasons for patient noncompliance: the
patient suffers adverse effects, the patient does not think the drug is effective, the
patient forgets to take the drug, the patient believes the disease is cured because
the symptoms have abated, the patient has misunderstood the user instructions,
the patient has run out of the drug, the patient does not master the administration
technique, e.g. inhalation, the drug formulation is unsuitable, the drug is unacceptable,
e.g. unpleasant taste, the patient uses many drugs simultaneously (polypharmacy),
frequent dosages and the patient has other objections towards the use of a certain
drug. In relation to drug therapy, the patient is compliant if he or she cooperates
fully in taking a prescribed medication following medical recommendations.Self- assessment 1.131. Why WHO recommended Direct Observed Therapy (DOT) for patient
taking anti-tuberculosis drugs?1.14. Therapeutic effectLearning activity 1.14 1. What do you observe on this image?
1. What do you observe on this image?
2. Discus the importance of taking the medication as prescribed?CONTENT SUMMARY
The main purpose of taking the medication is to achieve the therapeutic effect.
Therapeutic effect refers to the response after a treatment of any kind, the results
of which are judged to be desirable and beneficial. Therapeutic effects vary with the
nature of the medication, the length of time drugs was received and also vary with
client physical condition and interaction other drugs.The effect of a drug can be described at several levels: on the whole body, the
organ system(s), targets cell or at molecular target within cells.A single drug may have many effects other than its main therapeutic effect, and
in some instances these secondary effects and the responses they produce may
not be known in detail. Ideally, it is desirable that drugs should be as specific as
possible. This means that they should produce effects in as few organ systems as
possible – other than those in which an effect is required. The treatment can then
be controlled to achieve the desired effect.Drug response can be impacted by several factors including diet, comorbidities,
age, weight, drug–drug interactions, and genetics. Individual genetic variation in
key genes involved in the metabolism, transport, or drug target can contribute to
risk of adverse events or treatment failure.Self- assessment 1.141. Explain therapeutic effect?
2. Enumerate the factors affecting therapeutic effect of drug?textbook)1.15. Side effectsLearning activity 1.151. Read the book of pharmacology and explain side effects (using libraryCONTENT SUMMARYAn undesirable secondary effect which occurs in addition to the desired therapeutic
effect of a drug or medication is called side effect. It may vary for each individual
depending on the person’s disease state, age, weight, gender, ethnicity and general
health. All drugs have desirable or undesirable side effects. Even with a correct
drug dosage, side effects occur and are predicted.The terms side effects and adverse reactions are sometimes used interchangeably
in the literature and in speaking, but they are different. Some side effects are
expected as part of drug therapy. The occurrence of these expected but undesirableside effects is not a reason to discontinue therapy.
Side effects can occur when commencing, decreasing/increasing dosages, or
ending a drug or medication regimen. Side effects may also lead to non-compliance
with prescribed treatment. When side effects of a drug or medication are severe,
the dosage may be adjusted or a second medication may be prescribed. Lifestyle
or dietary changes may also help to minimize side effects.Self- assessment 1.151. What a nurse can do if the patient develops the side effect after
administrating the drug?2. Give an example where side effects may be desirable?1.16 Adverse reactionsLearning activity 1.16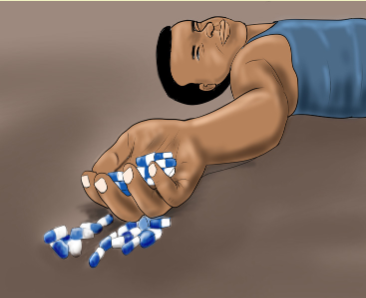 1. What do you observe on this image?
1. What do you observe on this image?
2. What do you thing happened to this person?
3. What can you do in this situation?CONTENT SUMMARYWhen the patient is taking the medications he/she can develop some effect which is
not desirable and severe which is called adverse effects. The adverse effects can
be classified into dose-related and non-dose-related effects.Dose-related adverse effects are associated with the drug’s known pharmacological
effects and occur when drugs are used in therapeutic doses. In principle, they are
predictable. All users will experience these adverse effects if the dose is high enough.
Often, an increase in the concentration of the drug due to reduced elimination, or
drug interactions which potentiate the effect, can be responsible for such adverse
effects. Toxic effects are included in this group.Non-Dose-Related Adverse Effects: In principle, all effects of drugs depend on
the dose that is taken (with a zero dose, there are no effects or adverse effects).
When adverse effects are classified as nondose- related, this means that such
effects occur at doses or concentrations that are considerably lower than the
standard dose known to produce a therapeutic effect. Such adverse effects are
not predictable, unless a patient has experienced them before. Typically, only a
few individuals experience non-dose-related adverse effects. Allergic reactions are
included in this group.Adverse reactions are more severe than side effects. The adverse reactions are
classified into type A (augmented) or type B (bizarre), C, D and E.Types A adverse reactions are therefore dose-dependent and predictable, but type
B Adverse reactions are neither predictable nor dose-dependent. Type A adverse
reactions are a kind of side effect and are normally associated with high morbidity
and low mortality. In contrast, type B adverse reactions are frequently severe or
bizarre reactions and are associated with low rates of morbidity but potentially high
rates of mortality. The immune system is commonly involved in type B adverse
reactions.Type A adverse reactions are more common than Type B reactions and account
for 80–90% of all reactions. They are a range of untoward effects (unintended and
occurring at normal doses) of drugs that cause mild to severe side effects, including
anaphylaxis (cardiovascular collapse). Adverse reactions are always undesirable.
Adverse effects must always be reported and documented because they represent
variances from planned therapy.Toxic effects, or toxicity is also an adverse drug reaction caused by excessive
dosing. However, for drugs that have a wide therapeutic index, the therapeutic
ranges are seldom given. For drugs with a narrow therapeutic index, such as
aminoglycoside antibiotics and anticonvulsants, the therapeutic ranges are closely
monitored. When the drug level exceeds the therapeutic range, toxic effects are
likely to occur from overdosing or drug accumulation.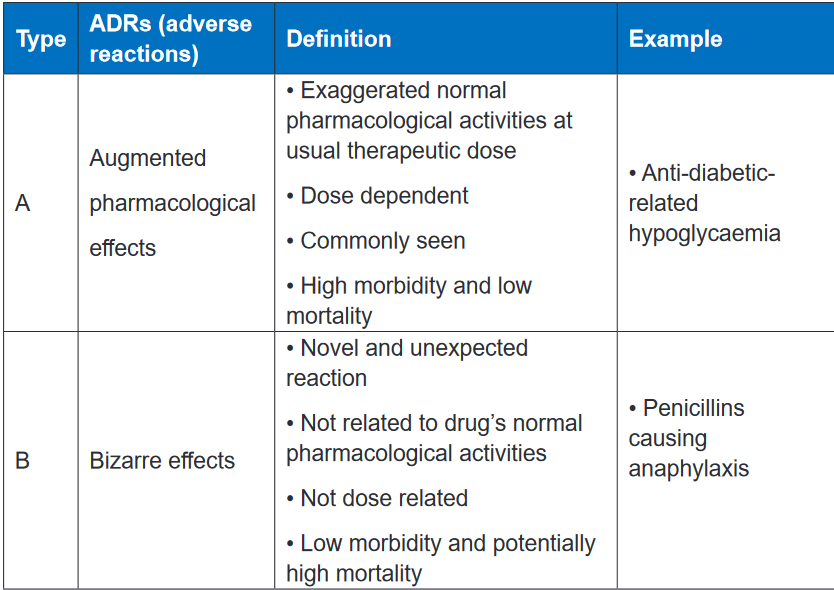 CONTENT SUMMARY
CONTENT SUMMARY
Effects most often manifest as changes in organ function. These may be changes in
the appearance of the skin, changes in the function of the respiratory, cardiovascular
and nervous systems, or changes in bone marrow function and the GI tract. The
liver and kidneys are particularly vulnerable, since the concentration of drugs and
their metabolites is usually high during drug elimination via these organs.The responsibility for reducing adverse reactions lies with everyone associated with
drug production and use. The pharmaceutical industry must strive to produce the
safest medicines possible; the prescriber must select the least harmful medicine
for a particular patient; the nurse must evaluate patients for adverse reactions and
educate patients in ways to avoid or minimize harm; and patients and their families
must watch for signs that an adverse reaction may be developing and should seek
medical attention if one appears.Anticipation of adverse reactions can help minimize them. Both the nurse and the
patient should know the major adverse reactions that a drug can produce. This
knowledge allows early identification of adverse effects, thereby permitting timely
implementation of measures to minimize harm. As noted, certain drugs are toxic to
specific organs. When patients are using these drugs, function of the target organ
should be monitored.The liver, kidneys, and bone marrow are important sites of drug toxicity. For drugs
that are toxic to the liver, the patient should be monitored for signs and symptoms of
liver damage (jaundice, dark urine, light-colored stools, nausea, vomiting, malaise,
abdominal discomfort, loss of appetite), and periodic Live Function Tests should
be performed. For drugs that are toxic to the kidneys, the patient should undergo
routine urinalysis and measurement of serum creatinine. In addition, periodic tests
of creatinine clearance should be performed. For drugs that are toxic to bone
marrow, periodic blood cell counts are required. Adverse effects can be reduced by
individualizing therapy.When choosing a drug for a particular patient, the prescriber must balance potential
risks of that drug versus its probable benefits. Drugs that are likely to harm a specific
patient should be avoided. For example, if a patient has a history of penicillin
allergy, we can avoid a potentially severe reaction by withholding penicillin and
administering a suitable substitute. Similarly, when treating pregnant patients, we
must withhold drugs that can injure the fetus.Lastly, we must be aware that patients with chronic disorders are especially
vulnerable to adverse reactions. In this group are patients with hypertension,
epilepsy, heart disease, and psychoses. When drugs must be used long term, the
patient should be informed about the adverse effects that may develop over time
and should be monitored for their appearance.Allergic Reaction is an immune response. For an allergic reaction to occur there
must be prior sensitization of the immune system. Once the immune system has
been sensitized to a drug, re-exposure to that drug can trigger an allergic response.
The intensity of allergic reactions can range from mild itching to severe rash to
anaphylaxis. Estimates suggest that less than 10% of adverse reactions are of the
allergic type.The intensity of an allergic reaction is determined primarily by the degree of
sensitization of the immune system not by drug dosage. Furthermore, since
a patient’s sensitivity to a drug can change over time, a dose that elicits a mild
reaction early in treatment may produce an intense reaction later on. In fact, most
serious reactions are caused by just one drug family, example the penicillins.The others drugs effect may include Paradoxical Effect, Iatrogenic Disease,
Carcinogenic Effect, Carcinogenic Effect, Teratogenic Effect, Organ-Specific
Toxicity and Hepatotoxic Drugs. Paradoxical Effect is the opposite of the intended
drug response. A common example is the insomnia and excitement that may occur
when some children and older adults are given benzodiazepines for sedation.
Iatrogenic Disease is a disease that occurs as the result of medical care or treatment.
The term iatrogenic disease is also used to denote a disease produced by drugs.Iatrogenic diseases are nearly identical to idiopathic (naturally occurring) diseases.
For example, patients taking certain antipsychotic drugs may develop a syndrome
whose symptoms closely resemble those of Parkinson disease. Carcinogenic Effect
refers to the ability of certain medications and environmental chemicals to cause
cancers.Teratogenic Effect is a drug and other chemicals capable of causing birth defects.
Organ-Specific Toxicity Many drugs are toxic to specific organs. Common examples
include injury to the kidneys caused by amphotericin B (an antifungal drug), injury
to the heart caused by doxorubicin (an anticancer drug), injury to the lungs caused
by amiodarone (an antidysrhythmic drug), and injury to the inner ear caused by
aminoglycoside antibiotics (e.g., gentamicin). Hepatotoxic Drugs as some drugs
undergo metabolism by the liver, they are converted to toxic products that can
injure liver cells. These drugs are called hepatotoxic drugs.However, many of these incidents are avoidable. Prescribers have to consider
the risk benefit ratio before prescribing a particular drug, and be aware that any
patient taking regular medication may develop an adverse reaction. Healthcare
professionals should know how to monitor, recognize and manage adverse drug
reactions or side effects, and this may involve stopping or changing the drug before
harm is done to patients.Self- assessment 1.161. What does the nurse have to do when the client develops allergic reaction
to the drugs?2. Discusses the concept of adverse drug reactions and drug side effects3. 30-year-old women client came to the health post where you work. She
is complaining hearing problem (tinnitus) 3 day ago after taking quinine.
How will you explain to the client about the symptom she developed after
taking quinine?4. Adverse drug reactions are mainly classified into reactions related to
the main pharmacological action of the drug (type A) and reactions that
are unpredictable and are not dose-related (Type B). Complete the table
below, using the key words and phrases provided in the box.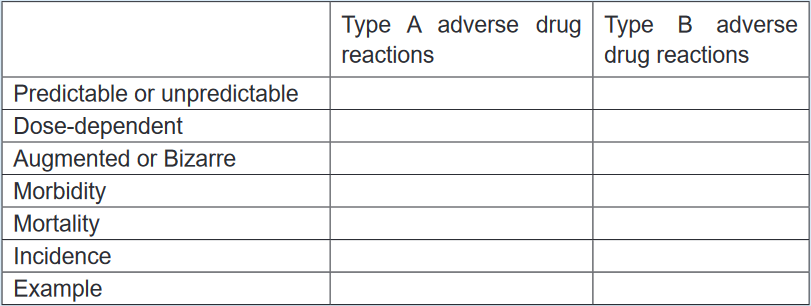 1.17 AntidotesLearning activity 1.171. Read the book of pharmacology and explain antidote (using library booksCONTENT SUMMARYDuring the drugs administration the patient can experience any unusual reaction
1.17 AntidotesLearning activity 1.171. Read the book of pharmacology and explain antidote (using library booksCONTENT SUMMARYDuring the drugs administration the patient can experience any unusual reaction
which can be reversed by administering another drug that acts as an antidote.
For example, when too much opiate is taken, the drug naloxone may be given to
counteract the effect. It is very important to monitor the drugs closely to detect or to
avoid any unusual reaction and have different antidotes at health facility which can
be used in case of overdose to prevent the complication associated. An antidote
is a drug, chelating substance, or a chemical that counteracts (neutralizes)
the effects of another drug or a poison.There are dozens of different antidotes; however, some may only counteract one
particular drug, whereas others (such as charcoal) may help reduce the toxicity of
numerous drugs. Antidotes mediate its effect either by preventing the absorption of
the toxin, by binding and neutralizing the poison, antagonizing its end-organ effect,
or by inhibition of conversion of the toxin to more toxic metabolites.Drug toxicity can be reversible or irreversible, depending on the organs involved.
Damage to the liver may be reversible, because liver cells can regenerate. However,
hearing loss from damage to the eighth cranial nerve caused by toxic reaction to the
anti-infective drug streptomycin may be permanent. Sometimes drug toxicity can
be reversed by administering another drug that acts as an antidote. For example,
when too much opiate is taken, naloxone may be given to counteract the effect.The FDA encourages nurses and other health care providers to report medication
errors to its database, which is used to assist other professionals in avoiding similar
mistakes. Poisoning occurs when an overdose of a drug damages multiple body
systems, leading to the potential for fatal reactions. Antidotes for drugs that can
cause potentially dangerous or fatal reactions must always be readily available.
Assessment parameters vary with the particular drug.Treatment of drug poisoning also varies, depending on the drug. Emergency and
life support measures often are needed in severe cases. Although some medication
errors go unreported, it is always the nurse’s legal and ethical responsibility to
document all occurrences. In severe cases, adverse reactions caused by medication
errors may require the initiation of lifesaving interventions for the patient, including
available antidotes. After such an incident, the patient may require close supervision,
and additional medical treatments may be warranted.According to mode of action the antidote can be classified as physical, chemical
and physiological and pharmacological. Physical antidote the agent use to interfere
with poison through physical properties by adsorbing. Chemical antidote interacts
specifically with a toxicant, or neutralize the toxicant. Physiological antidote act by
producing opposite effect to that of poison. Pharmacological antidote counteracts
the effects of a poison by producing the opposite pharmacological effects. They
may neutralize or antagonize the effects of a toxicant.According to site of action the antidote may act by preventing the formation of toxic
metabolites, by facilitation of more rapid or complete elimination of toxicant, and by
competing with the toxicant’s action at a receptor site. For preventing the formation
of toxic metabolites: more effective when given immediately before toxic metabolic
activation. For facilitation of more rapid or complete elimination of toxicant: change
the physiochemical nature of toxin, allowing better glomerular filtration and prohibit
tubular reabsorption.Self- assessment 1.171. Explain the mechanism of action of the antidote?
2. Give example of drug toxicity which can be reversible or irreversible,
depending on the organs involved1.18 Responsibilities of nurses regarding safe drug
administrationLearning activity 1.181. What is the role of nurse and responsibilities in medication administration?CONTENT SUMMARYNurses have a unique role and responsibility in medication administration, in that
they are frequently the final person to check to see that the medication is correctly
prescribed and dispensed before administration. It is standard during nursing
education to receive instruction on a guide to clinical medication administration
and upholding patient safety known as the ‘Nine rights’ or ‘Nine R’s’ of medication
administration (Right Patient, Right Reason or Indication, Right drug, Right dose,
Right Route and form, Right Time, Right Documentation, Right Response and Right
to Refuse).Right Patient, when administering a drug, it is important to use two methods
(visual as well as verbal methods) to identify the patient before administering the
medication. Nurse must be certain that the patient receiving the drug is the patient
for whom the drug has been ordered by reading properly the physician’s order. Call
the patient by name and ask him to repeat his name aloud. Be very careful if the
patient is deaf or otherwise does not understand the language.A visual identifier may include checking the patient’s name on his or her wristband,
on the patient’s card and on the medicine card for matching name and ID number
as on a chart. It is advisable not to address patients by first name or surname
alone, in the event, there are two or more patients with identical or similar names
in a unit. Depending on the unit that a patient may be in, some patients, such as
psychiatric patients, may not wear wristbands or may have altered mentation to the
point where they are unable to identify themselves correctly. In these instances,
nurses are advised to confirm a patient’s identity through alternative means with
appropriate due diligence.If there is no written identification verifying the patient’s name, nurse should obtain
a wristband or other form of identification before administering the drug. Nursemay also ask the patient to identify him- or herself and request another unique
identifier such as date of birth. However, do not ask, “Are you Mr or Mrs A?” Some
patients, particularly those who are confused or have difficulty hearing, may respond
by answering “yes” even though that is not their name. Some long-term care or
rehabilitation care facilities have pictures of the patient available, which allow the
nurse to verify the correct patient. If pictures are used to identify patients, it is critical
that they are recent and bear a good likeness of the individual.Right Reason or Indication addresses the appropriateness in use of the medication
to the patient. Confirm the rationale for use through researching the patient’s history
while also asking the patient the reason he or she is taking the drug. Always revisit
the rationale for long-term medication use. Knowledge of the drug’s indication
allows the nurse, prescriber, members of the health care team, patient and/or family
members to understand what is being treated. Understanding the indication helps
pharmacists and nurses to catch potential errors, provide thorough explanations to
the patient/family, and decrease challenges to medication reconciliation.The nurse has the responsibility to verify the reason that the patient is receiving
the medication. It is important to understand the indication, which is related to the
medical diagnosis. If in doubt about the reason for the order, the nurse must verify
the medication order with the prescriber before administration.Right medication or drug, some brand names or generic names may have very
similar spelling or sound very similar due to prefix, suffix, or starting with the same
first letter. Poor handwriting and abbreviations account for many medical errors
due to misreading letters or numerals that appear differently to different individuals.
Right Drug names can be confused, especially when the names sound similar, or
the spellings are similar.Quickly preparing a drug for administration or failing to look up questionable drugs
can put you at increased risk for administering the wrong drug. An error in drug
name or amount can be found when nurse compares the medication administration
record: with the container label, as the item is removed from the card, and before
the actual administration of the drug.The nurse must be careful of drugs whose names sound alike. When administering
medications, the nurse compares the label of the medication container with the
medication form three times: before removing the container from the drawer or
shelf, as the amount of medication ordered is removed from the container and
before returning the container to storage.The nurse must look for colour, odour, and consistency of the drug. Unusual
characteristics of the drugs should be questioned. The nurse must also administer
medicine only from clearly labelled container and remember to check other critical
information on packaging such as the expiration date.The nursing providers should also develop a routine habit of explicitly asking patients
about known allergies or history of an allergic. The conversation or anything that
distracts the mind not recommended during drug administration. The nurse must be
familiar with the trade names.If there is doubt consult the physician or at least seniors or other reliable sources.
Avoid accepting the verbal orders, only in emergencies are accepted. Always
identify the patient before giving medication. The nurse must make sure that the
drug has not been discontinued by the prescriber.The nurses administer only the medications they prepare. If an error occurs, the
nurse who administers the medication is responsible for the error. Clients who
self-administer medications should keep them in their original labelled containers,
separate from other medications, to avoid confusion.Right Route and form, a nurse must know the particulars about each medication
before administering it to ensure that the right drug, dose, route, and dosage form
are being used. A complete medication order includes the route of administration.
Confirm the appropriateness of the prescribed route while also making sure the
patient can take/receive the medication by the prescribed route. If a medication
order does not include the route, be sure to ask the prescriber to clarify it. Never
assume the route of administration.In addition, it is critical to patient safety to be aware of the right form of medication.
For example, there are various dosage forms of a commonly used medication,
acetaminophen.It is available in oral suspension, tablet, capsule, gelcap, and pediatric drops, as
well as rectal suppository dosage forms. Nurses need to give the right drug via the
right route with use of the correct dosage form.Medications can be given to patients in different many ways, all of which vary in the
time it takes to absorb the chemical, time it takes for the drug to act, and potential
side-effects based on the mode of administrations, include oral, intramuscular,
intravenous, topical, or subcutaneous injection and others. It is crucial that
nurses remain educated and up to date on newer medications or less commonly
administered medications to learn how they are safely delivered to patients before
being asked to do so in clinical practice.If a prescriber’s order does not designate a route of administration, the nurse
consults the prescriber. The nurse should alert the prescriber immediately if the
specified route is not the recommended route and he/she must report immediately
if an error occurs in the medication. The nurse must know and must be familiar with
the abbreviations used to designate the route of administration.‘Right time’, medications can be given to patients in different many ways, all of
which vary in the time it takes to absorb the chemical, time it takes for the drug
to act, and potential side-effects. Certain drugs have specific intervals or window-
periods during which another dose should be given to maintain a therapeutic effect
or level.Often, a guiding principle of this ‘right’ is that medications should be prescribed as
closely to the time as possible, and nurses should not deviate from this time by
more than half an hour to avoid consequences such as altering bioavailability or
other chemical mechanisms. Similarly, it is crucial that medications that are given
by an infusion, such as intravenous medications, are administered at the correct
rate.Failure to deliver a drug at the correct rate may lead to devastating consequences
for a patient. For example, vancomycin requires administration by slow intravenous
infusion to avoid a complication known as “red man syndrome,” a hypersensitivity
reaction that is managed by further slowing the infusion rate of vancomycin or
discontinuing the agent altogether.The administering medications at a time that was intended by the prescriber. The
nurse must Read the physician’s orders, know the hospital routines for the interval,
know the abbreviations for the time, give the medicine near the time ordered,
give the medicine as ordered in relation to the food intake and give the medicines
according to the actions expected. E.g., sleeping pills are given at bedtime.‘Right dose’, incorrect dosage, conversion of units, and incorrect substance
concentration are a prevalent modality of medication administration error. This error
type stems from nurses giving a patient an incorrect dose of medications, even if it
is the correct medication and the patient’s identity is verified, without first checking
to ensure it is the correct strength for the patient. This error type may be due to
misplaced decimals, errors in arithmetic, or incorrect conversion between two units.The nurse must have adapted observing positive behaviors to reduce medical
errors include consulting with pharmacy personnel, read physician orders to know
the correct dose, consider the age and weight of the patient, know the minimum
and maximum dose of the medicine administered, using calculators to assist in
arithmetic, or in some cases, cross-consulting with patients or their families about
usual doses they administer at home. Use ounce glasses instead of teaspoons to
measure ounces accurately, have written order before you prepare the drug, avoid
conversation or anything that distracts the mind.Right Documentation, medication error can result from inaccurate documentation.
Nurse should ensure appropriate documentations clearly reflect the client’s name,
the name of the ordered medications, the time the medication was administered, the
medication’s dosage, route, the date or the method of administration, frequency, thesignature of the physician, and Standing orders or routine medication orders. If any
of this information is missing the nurse should verify the order with the prescriber.After the administration of any drug, record the process immediately. Immediate
documentation is particularly important when drugs are given on an as-needed
(PRN) basis. For example, most analgesics require 20 to 30 minutes before the
drug begins to relieve pain.A patient may forget that he or she received a drug for pain, may not understand
that the administered drug was for pain, or may not know that pain relief is not
immediate, and may ask another nurse for the drug again. If the administration
of the analgesic was not recorded, the patient might receive a second dose of
the analgesic shortly after the first dose. This type of situation can be extremely
serious, especially when opioids or other central nervous system depressants are
administered. Immediate documentation prevents accidental administration of a
drug by another individual and it is essential to the process of administering drugs
correctly.Right Response refers to the drug and its desired response in the patient.
Continually assess and evaluate the achievement of the desired response, as
well as any undesired response. Examples of data gathering include, but are not
limited to, monitoring vital signs, weight, edema, intake and output, nutritional
intake, laboratory values, results of diagnostic testing, and auscultating heart and
lung sounds. Document any assessment, intervention, and monitoring as deemed
appropriate.Right to Refuse, the ninth right is that of the right of the patient to refuse. Patients
refuse medications for a variety of reasons. If refusal of a medication occurs, always
respect the patient’s right (to refuse), determine the reason, and take appropriate
action, including notifying the prescriber. Do not force! Document the refusal and
a concise description of the reason for refusal. Document any further actions you
take at this time, such as vital signs and/or system assessment.If a consequence to the patient’s condition and/or as hospital policy dictates, the
prescriber is to be contacted immediately. Never return unwrapped medication to a
container, and discard medication dose according to agency policy. If the wrapper
remains intact, return the medication to the automated medication-dispensing
system. Revise the nursing care plan as needed.The nurses bear the sole responsibility and accountability while administering
drug. They are responsible for their actions regardless of whether there is a written
order. If the physician’s prescription appears unreasonable or wrong, the nurses
should clarify with the doctor who prescribed the drug and get it clarified before
administering.The primary health care provider’s order must include the patient’s name, the drug
name, the dosage form and route, the dosage to be administered, the frequency
of administration and the health care provider’s signature and must follow the drug
order. In an emergency, nurse may administer a drug with a verbal order from the
primary health care provider. However, the primary health care provider must write
and sign the order as soon as the emergency is over. If a verbal order is given over
the telephone, write down the order, repeat back the information exactly as written,
and then ask for a verbal confirmation that it is correct. Any order that is unclear
should be questioned, particularly unclear directions for the administration of the
drug or a drug dose that is higher or lower than the dosages given in approved
references.Ways to prevent medication administration errors:• Read the medication labels carefully many products come in similar containers,
colors and shapes.• Be aware of medications with similar names many medication names sound
alike.• When new or unfamiliar medication is ordered, consult resource if prescriber
is also unfamiliar with drug, there is greater risk of inaccurate dosages being
ordered.• Do not administer medication ordered by nickname or unofficial abbreviation
know client with same last names.• Do not confuse equivalents.• Client should be educated regarding the self-administration of drugs while
getting discharged.Drug administration is a fundamental nursing responsibility. By understanding the
basic concepts of administering drugs safely and accurately, monitoring of the
therapeutic response (desired response) and reporting adverse reactions is critical.
Additionally, in the ambulatory setting, nurses are responsible for teaching the
patient and family members the information needed to self-administer drugs safely
in the home.Self- assessment 1.181. Describes the six right of drug administration?1.19. Food and drug administration (FDA) pregnancy risk
categoriesLearning activity 1.191. Read the book of pharmacology and explain why pregnant women cannot
take any drugs without medical prescription (using library textbook).CONTENT SUMMARYDrugs used by pregnant women may reach the fetus through the placenta and lead
to effects on the development, intellectual ability, birth defects, miscarriage and
stillbirth. The Food and Drug Administration has established five categories (A, B,
C, D &X) to indicate the potential for a systemically absorbed drug to cause birth
defects. The key differentiation among the categoriesCategory A: Adequate and well-controlled studies have failed to demonstrate
a risk to the fetus in the first trimester of pregnancy (and there is no evidence of
risk in later trimesters). Example drugs or substances: levothyroxine, folic acid,
liothyronine.Category B: Animal reproduction studies have failed to demonstrate a risk to the
fetus and there are no adequate and well-controlled studies in pregnant women.
Example drugs: metformin, hydrochlorothiazide, cyclobenzaprine, amoxicillin.Category C: Animal reproduction studies have shown an adverse effect on the
fetus and there are no adequate and well-controlled studies in humans, but potential
benefits may warrant use of the drug in pregnant women despite potential risks.
Example drugs: gabapentin, amlodipine, trazodone.Category D: There is positive evidence of human fetal risk based on adverse
reaction data from investigational or marketing experience or studies in humans,
but potential benefits may warrant use of the drug in pregnant women despite
potential risks. Example drugs: losartan.Category X: Studies in animals or humans have demonstrated fetal abnormalities
and/or there is positive evidence of human fetal risk based on adverse reaction
data from investigational or marketing experience, and the risks involved in use
of the drug in pregnant women clearly outweigh potential benefits. Example
drugs: atorvastatin, simvastatin, methotrexate, finasterideRegardless of the designated Pregnancy Category or presumed safety, no drug
should be administered during pregnancy unless it is clearly needed.Self- assessment 1.191. Describe FDA pregnancy risk categories?1.20 End unit assessmentEnd Unit assessment 11. Define pharmacology2. List 4 drugs dosage forms
3. What are the sources of drug?
4. Give the difference between loading dose and maintenance dose5. The nurse knows the importance of administering the right medication to
the patient and that drugs have many names. It is therefore important that
drugs be ordered by which name?6. Explain the importance of directly observed therapy in patient care
7. Explain the importance of fixed dose combination in patient care
8. How medication administration errors can be prevented?
9. What are the responsibilities of the nurse during drug administration?10. Drug X is given a prescription that reads as follows: “Ferrous sulfate 325
mg, PO for anemia.” When she goes to the pharmacy, the pharmacist
tells her that the prescription is incomplete. What is missing? What should
be done?11. Briefly discuss the “Nine Rights” and other “Rights” associated with safe
medication administration12. Clinical pharmacology is the study of
A. The biological effects of chemicals.B. Drugs used to treat, prevent, or diagnose disease.
C. Plant components that can be used as medicines.
D. Binders and other vehicles for delivering medication.13. The generic name of a drug is
A. The name assigned to the drug by the pharmaceutical company
developing it.
B. The chemical name of the drug based on its chemical structure.C. The original name assigned to the drug at the beginning of the evaluation
process.
D. The name that is often used in advertising campaigns.14. The Food and Drug Administration (FDA) pregnancy categories
A. Indicate a drug’s potential or actual teratogenic effects.
B. Are used for research purposes only.
C. List drugs that are more likely to have addicting properties.
D. Are tightly regulated by the Drug Enforcement Agency (DEA).15. Give the definition for a therapeutic dose:
A. The amount of a substance to produce the minimal biological effect
B. The amount of a substance to produce effects hazardous for an organism
C. The amount of a substance to produce the required effect in most
patients
D. The amount of a substance to accelerate an increase of concentration
of medicine in an organism16. Pick out the correct definition of a toxic dose:
A. The amount of substance to produce the minimal biological effect
B. The amount of substance to produce effects hazardous for an organism
C. The amount of substance to produce the necessary effect in most of
patients
D. The amount of substance to fast creation of high concentration of
medicine in an organism17. A rectal suppository is used to treat fever. This would represent what type
of drug delivery?
A. Parenteral and local
B. Parenteral and systemic
C. Enteral and local
D. Enteral and systemic18. Which of the following is not a semisolid dosage forms?
A. Solution
B. CreamC. Paste
D. Gel19. A suppository is generally intended for use in
A. Rectum
B. Ear
C. Nose
D. Mouth20. Vaginal suppositories also called as
A. Simple suppositories
B. Bougies
C. Pessaries
D. Soft tablet21. The nurse is reviewing the various forms of topical medications. Which of
these are considered topical medications?
A. Tablets for oral route
B. Eye drops for inflammation
C. Sublingual tablet for chest pain
D. Intradermal injection for tuberculosis testing22. An 82-year-old patient is admitted to the hospital after an episode of
confusion at home. The nurse is assessing the current medications he
is taking at home. Which method is the best way to assess his home
medications?
A. Ask the patient what medications he takes at home.
B. Ask the patient’s wife what medications he takes at home.
C. Ask the patient’s wife to bring his medications to the hospital in their
original containers.
D. Contact the patient’s pharmacy for a list of the patient’s current
medications.23. During the medication administration process, it is important that the
nurse remembers which guideline?
A. When in doubt about a drug, ask a colleague about it before giving the
drug.
B. Ask what the patient knows about the drug before giving it.C. When giving a new drug, be sure to read about it after giving it.
D. If a patient expresses a concern about a drug, stop, listen, and
investigate the concerns.24. A patient’s medical record includes an order that reads as follows:
“Pacetamol 500 mg once daily at 09h00.” Which action by the nurse is
correct?
A. The nurse does not give the drug.
B. The nurse gives the drug orally.
C. The nurse gives the drug intravenously.
D. The nurse contacts the prescriber to clarify the dosage route.
