UNIT 12: IONIC AND METALLIC BONDS
Key unit competence
Describe how properties of ionic compounds and metals are related to the
nature of their bonding.
Introductory ActivityConsider the following figure 12.1 below.
The figure shows materials commonly used at home. If you reflect back
around your house/home you will see hundreds of objects made from
different kinds of materials.
a) Observe the objects (in picture) and classify them according to the
materials they are made of.
b) Have you ever wondered why the manufacturers choose the
material they did for each item?
c) Why are frying saucepans made of metals and dishes, cups and
plates often made of glass and ceramic?
d) Could dishes be made of metal? And saucepans made of ceramic
and glass?
Of the total number of individual chemically pure substances known, several
million are compounds, formed when two or more elements are chemically
bonded together and less than 100 are elements. Only six of these elements
consist of free, unbounded atoms at room temperature. These are the noble
gases. All other elements exist as individual or giant molecules, or metallic
lattices, in which atoms are chemically bonded to each other.
12.1. Explanations of why atoms of elements form bonds
Activity 12.1
Consider Chlorine (Cl, Z = 17) and Argon (Ar, 18) atoms of the elements of
Period 3 in the Periodic Table.
a) Which of these atoms is more reactive?
b) Suggest the reasons for your answer in (a) in terms of the
electronegativity and electronic structure.
c) Choose, between Chlorine and Argon, which one has lower energy
potential.
The atoms of most elements form chemical bonds because the atoms become
more stable when bonded together. Electric forces attract neighbouring
atoms to each other, making them stick together.
In atoms, electrons are arranged into complex layers called shells. For most
atoms, the outermost shell is incomplete, and the atom shares electrons with
other atoms to fill the shell.
The type of chemical bond maximizes the stability of the atoms that form it.
An ionic bond, where one atom essentially donates an electron to another,
forms when one atom becomes stable by losing its outer electrons and the
other atoms become stable (usually by filling its valence shell) by gaining the
electrons. Covalent bonds form when sharing atoms results in the highest
stability.
Other types of bonds https://www.thoughtco.com/types-of-chemical-bonds-
603984 besides ionic and covalent chemical bonds exist, too.
Atoms with incomplete shells are said to have high potential energy; atoms
whose outer shells are full have low potential energy. In nature, objects with
high potential energy “seek” a lower energy, becoming more stable as a
result. Atoms form chemical bonds to achieve lower potential energy.
Application activity 12.1
Explain why atoms of elements form bonds in terms of stability and potential
energy.
12.2. Gain of stability by losing and gaining electrons
Activity 12.2
The figure below shows how the atoms may gain stability by losing andgaining electrons.
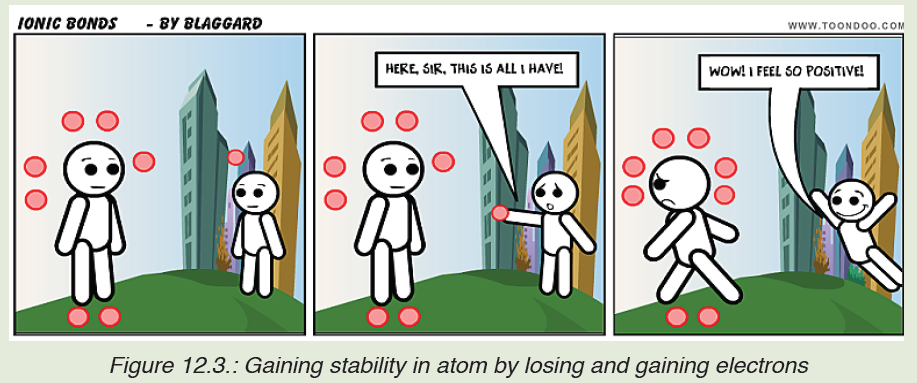
d) State and describe clearly the group on the periodic table that
contains elements that have the same atomic structures as the
species obtained when atom “Big” has received the electron from
atom “Little”.
Like people always relate and connect to others depending on their values,
interests and goals so do unstable atoms. They combine together to achieve
stability. We know that noble gases are the most stable elements in the
periodic table. They have a filled outer electron energy level.
When an atom loses, gains, or shares electrons through bonding to achieve
a filled outer electron energy level, the resulting compound is often more
stable than individual separate atoms.
• Neutral sodium has one valence electron. When it loses this electron
to chlorine, the resulting Na+ cation has an outermost electron energy
level that contains eight electrons. It is isoelectronic (same electronic
configuration) with the noble gas neon.
• On the other hand, chlorine has an outer electron energy level that
contains seven electrons. When chlorine gains sodium’s electron, it
becomes an anion that is isoelectronic with the noble gas argon.
It is easiest to apply the “Octet Rule” to predict whether two atoms will form
bonds and how many bonds they will form. Most atoms need 8 electrons
to complete their outer shell. So, an atom that has 2 outer electrons will
often form a chemical bond with an atom that lacks two electrons to be
“complete”. The octet rule states that elements gain or lose electrons to
attain an electron configuration of the nearest noble gas. Octet comes from
Latin language meaning “eight”.
Note that the “Duet Rule” is also applied. The noble gas HELIUM has two
electrons (a doublet) in its outer shell, which is very stable. Hydrogen only
needs one additional electron to attain this stable configuration, while lithium
needs to lose one.
Low atomic weight elements (the first twenty elements) are most likely to
adhere to the Octet Rule. For example,
• A sodium atom has one lone electron in its outer shell.
• A chlorine atom, in contrast, is short one electron to fill its outer shell.
• Sodium readily donates (loses) its outer electron (forming the
Na+ ion, since it then has one more proton than it has electrons), while
chlorine readily accepts (gains) a donated electrons (making the Cl- ion, since chlorine is stable when it has one more electron than it
has protons).
• Sodium and chlorine form an ionic bond with each other, to form
table salt or sodium chloride.
Application activity 12.2
1) State the following Rule?
a. Octet Rule
b. Duet Rule
2) Answer to the following questions
a. Does sodium need to gain electron than chlorine? (Yes or No)
b. Explain the target of sodium when it is seeking to lose electron and
chlorine to gain electron.
3) Which of the following is stable? Explain why?
i. Na+ iii. Clii. Na iv. Cl-
12.3. Ionic bonding
Activity 12.3
In the Ordinary Level, you learnt that there exist three main types of
chemical bonding namely, covalent, ionic and metallic.
a) Recall the definition of the ionic bond.
b) The bond formed between sodium and chlorine atoms to form the
table salt is an ionic bond. Using experiments performed in daily life
and performing the non-performed ones, state the properties of a
table salt and use it to generalize the properties of ionic compounds
basing on the criteria given in parentheses (Appearance/physical
state, Solubility in water and in organic solvent, Temperature
required to melt, Electrical conductivity of solid and aqueous
solution).
An ionic bond is a chemical bond formed between two ions with opposite
charges. Ionic bonds form when one atom gives up one or more electrons
to another atom. These bonds can form between a pair of atoms or between
molecules and are the type of bond found in salts.
12.3.1. Ionic bond formation
Once the oppositely charged ions formed when electrons are transferred
from one atom to another, they are attracted by their positive and negative
charges (by electrostatic forces) and form an ionic compound. Ionic bonds
are also formed when there is a large electronegativity difference between
two atoms. This difference causes an unequal sharing of electrons such that
one atom completely loses one or more electrons and the other atom
gains one or more electrons. For example, in the creation of an ionic bond
between a metal atom, sodium (electronegativity = 0.93) and a non-metal,
fluorine (electronegativity = 3.98).Let us take a look of how sodium and fluorine bond to form sodium fluoride:
The curved arrow between sodium and fluorine atoms represents the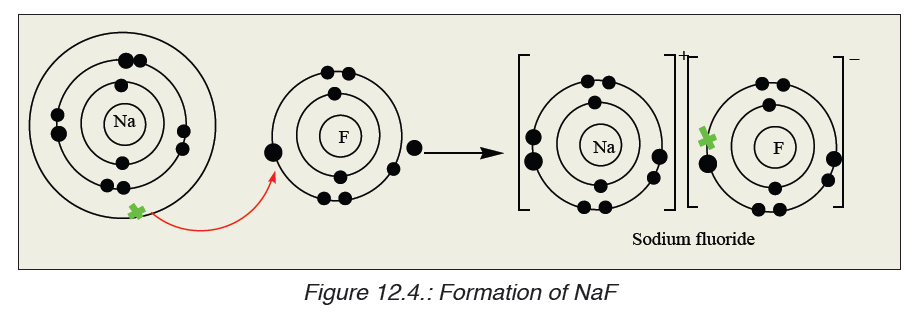
transfer of an electron from a sodium atom to a fluorine atom to form
oppositely charged ions. These two ions are strongly attracted to each
other because of their opposite charges. A bond is now formed and the
resulting compound is called Sodium fluoride.
Another example of ionic bonding formation is the formation of magnesiumoxide.
12.3.2. Properties of ionic compounds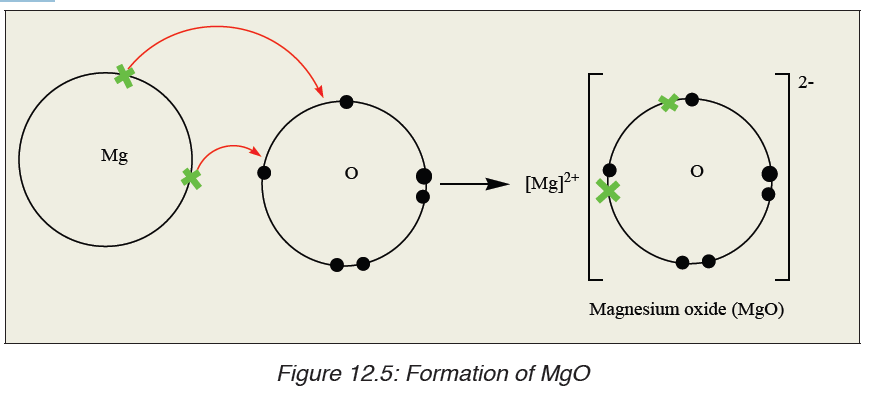
Here are the properties shared by the ionic compounds. Notice that the
properties of ionic compounds relate to how strongly the positive and
negative ions attract each other in an ionic bond.
1. They have high melting points and high boiling points
In an ionic lattice, there are many strong electrostatic attractions between
oppositely charged ions. We therefore expect that ionic solids will have
high melting points. On melting although the regular lattice is broken down,
there will still be significant attractions between the ions in the liquid. This
should result in high boiling points also.
The factors which affect the melting point of an ionic compound are:
• The charge on the ions: «The greater the charge, the greater the
electrostatic attraction, the stronger the ionic bond, the higher the
melting point ». For example, Melting Point of NaCl is 801 oC and that
of MgO is 2,800 oC.
• The size of the ions: «Smaller ions can pack closer together than
larger ions so the electrostatic attraction is greater, the ionic bond is
stronger, the melting point is higher». For example, Melting Point of
NaF is 992 oC and that of CsF is 2,800 oC.
2. Most ionic compounds are soluble in water
This is because the electrostatic forces of the polar water molecules are
stronger than the electrostatic forces keeping the ions together. When
an ionic compound like NaCl is added to water, water molecules attract the
positive and negative salt ions. Water molecules surround each ion and move
the ions apart from each other. The separated ions dissolve in water. There
are several exceptions, however, where the electrostatic forces between the
ions in an ionic compound are strong enough that the water molecules cannot
separate them. Despite these few limitations, water›s ability to dissolve ionic
compounds is one of the major reasons it is so vital to life on Earth. Ionic
compounds are generally insoluble in non-polar solvents like kerosene.
3. They are hard and brittle
Ionic crystals are hard because the positive and negative ions are strongly
attracted to each other and difficult to separate, however, ionic solids are
brittle. When a stress is applied to the ionic lattice, the layers shift slightly.
The layers are arranged so that each cation is surrounded by anions in
the lattice. If the layers shift then ions of the same charge will be brought
closer together. Ions of the same charge will repel each other, so the latticestructure breaks down into smaller pieces.
4. They conduct electricity when molten or dissolved in water
In order for a substance to conduct electricity, it must contain mobile
particles capable of carrying charge.
• Solid ionic compounds do not conduct electricity because the ions
(charged particles) are locked into a rigid lattice or array. The ions
cannot move out of the lattice, so the solid cannot conduct electricity.
• When is molten, the ions are free to move out of the lattice structure.
– Cations (positive ions) move towards the negative electrode (cathode):
M+ + e- → M
– Anions (negative ions) move towards the positive electrode (anode):
X- → X + e-
• When is dissolved in water to form an aqueous solution, the ions are
released from the lattice structure and are free to move so the solution
conducts electricity just like the molten (liquid) ionic compound.
5. They form crystals
Ionic compounds form crystal lattices rather than amorphous solids. Although
molecular compounds form crystals, they frequently take other forms but
molecular crystals typically are softer than ionic crystals. At an atomic
level, an ionic crystal is a regular structure, with the cation and anion
alternating with each other and forming a three-dimensional structure
based largely on the smaller ion evenly filling in the gaps between thelarger ion.
Application activity 12.3
1. The diagram below represents a part of the structure of sodium
chloride. The ionic charge is shown on the centre of only one ofthe ions.
a) On the diagram, mark the charges on the four negative ions.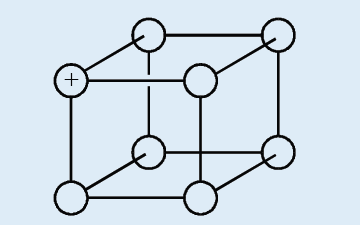
b) What change occurs to the motion of the ions in sodium chloride
when it is heated from room temperature to a temperature below
its melting point?
c) Sodium chloride can be formed by reacting sodium with chlorine.
A chloride ion has one more electron than a chlorine atom. In the
formation of sodium chloride, from where does this electron come?
2. Draw diagrams to illustrate the formation of ionic compounds in the
following substances:
a) Magnesium chloride
b) Sodium peroxide
c) Iron (III) chloride
d) Sodium sulphide
3. Solid sodium chloride and solid magnesium oxide are both held
together by ionic (electrovalent) bonds.
a) Using s, p, d and f notation, write down the symbol for and the
electronic configuration of (i) a sodium ion; (ii) a chloride ion; (iii) a
magnesium ion; (iv) an oxide ion.
b) Explain what holds sodium and chloride ions together in the solid
crystal
c) Sodium chloride melts at 1074 K; magnesium oxide melts at 3125
K. Both have identical structures. Why is there such a difference intheir melting points?
12.4. Metallic bonding
Activity 12.4
1. Give three examples of substances which are malleable, ductile,
good conductor of heat and electricity, and having a characteristic
luster. Here you can use a dictionary or other searching tools to
find the meaning for any unfamiliar word.
2. Suggest another property, apart from those given, of the substances
you have given in 1.
3. Choose from the examples given in 1, one which is most common
and well known.
a) This substance is seen to be composed by atoms of one element.
Which one?
b) Use a labelled drawing to show the internal structure of that kind of
substance.
A metallic bond is a type of chemical bond formed between positively
charged atoms in which the free electrons are shared among a lattice
of cations. In contrast, covalent and ionic bonds form between two discrete
(separate) atoms.
Metallic bonding is the main type of chemical bonds that forms between
metal atoms (pure metals and alloys and some metalloids). A metal is alattice of positive metal “ions” in a “sea” of delocalised electrons.
12.4.1. Formation of metallic bond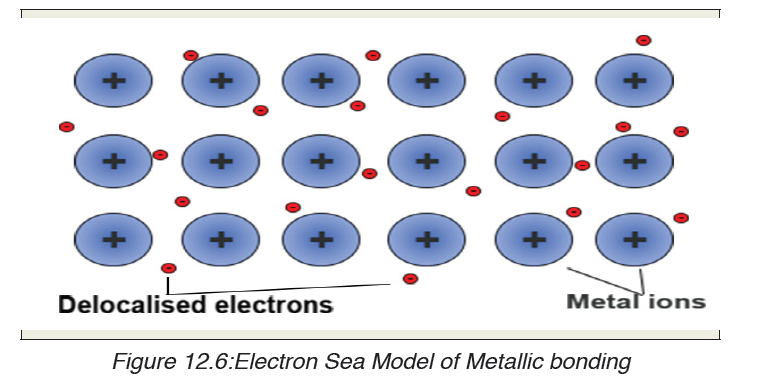
The outer energy levels of metal atoms (the s and p orbitals) overlap. At least
one of the valence electrons participating in a metallic bond is not shared
with a neighbour atom, nor is it lost to form an ion. Instead, the electrons
form what may be termed an «electron sea” in which valence electrons
are free to move from one atom to another. Metallic bonding refers to theinteraction between the delocalised electrons and the metal nuclei.
The electron sea model is an oversimplification of metallic bonding.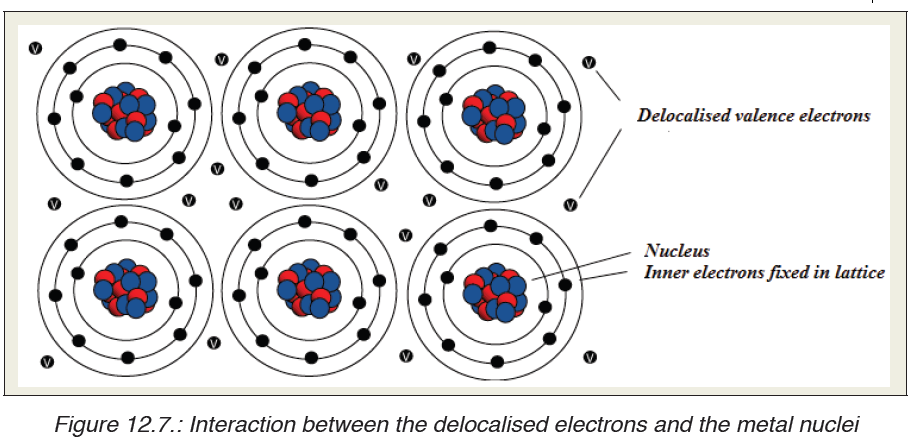
Calculations based on electronic band structure or density functions are
more accurate. Metallic bonding may be seen as a consequence of a
material having many more delocalized energy states than it has delocalized
electrons (electron deficiency), so localized unpaired electrons may become
delocalized and mobile. The electrons can change energy states and move
throughout a lattice in any direction.
Bonding can also take the form of metallic cluster formation, in which
delocalized electrons flow around localized cores. Bond formation depends
heavily on conditions. For example, hydrogen is a metal under high pressure!
As pressure is reduced, bonding changes from metallic to non-polar covalent.
12.4.2. Physical properties of metals
Because electrons are delocalized around positively charged nuclei, metallic
bonding explains many properties of metals. The three main factors that
affect the strength of a metallic bond are:
– Number of protons/ Strength of nuclear attraction: The more
protons the stronger the force of attraction between the positive ions
and the delocalized electrons
– Number of delocalized electrons per atom: The more delocalized
electrons the stronger the force of attraction between the positive ions
and the delocalized electrons
– Size of atom: The smaller the atom, the stronger the force of attraction
between the positive ions and the delocalized electrons and vice-versa,
the larger the atom, the weaker the force of attraction between the
positive ions and the delocalised electrons.
The main physical properties of metallic metals are given below.
1. Electrical Conductivity
Most metals are excellent electrical conductors because the electrons
in the electron sea are free to move and carry charge. For example,
electric wires in our homes are made of aluminium and copper. They are
good conductor of electricity. Electricity flows most easily through gold,
silver, copper and aluminium. Gold and silver are used for fine electrical
contacts in computers.
2. Thermal Conductivity
Metals conduct heat because the free electrons are able to transfer
energy away from the heat source and also because vibrations of atoms
(phonons) move through a solid metal as a wave. Cooking utensils and
water boilers are also made of iron, copper and aluminium, because they aregood conductors of heat.
3. Ductility
Metals tend to be ductile or able to be drawn into thin wires because
local bonds between atoms can be easily broken and also reformed.
Single atoms or entire sheets of them can slide past each other and reform
bonds. Wires are mainly made from copper, aluminium, iron and magnesium.
4. Malleability
Metals are often malleable or capable of being molded or pounded
into a shape, again because bonds between atoms readily break and
reform. This ability to bend or be shaped without breaking occurs because
the electrons simply slide over each other instead of separating. The
binding force between metals is non-directional, so drawing or shaping a
metal is less likely to fracture it. Electrons in a crystal may be replaced
by others. Gold and Silver metals are the most malleable metals. They can
be hammered into very fine sheets. Thin aluminium foils are widely used for
safe wrapping of medicines, chocolates and food material.
5. Metallic Luster
Metals tend to be shiny or display metallic luster. They are opaque
once a certain minimum thickness is achieved. The electron sea reflects
photons off the smooth surface therefore there is an upper frequency limit
to the light that can be reflected. Silver is a very good reflector. It reflects
about 90% of the light falling on it. All modern mirrors contain a thin coating
of metals. Due to their shiny appearance they can be used in jewellery and
decorations.
Application activity 12.4
1. Magnesium has a higher melting and boiling point than sodium. This can
be explained in terms of the electronic structures, the packing, and the
atomic radii of the two elements.
a) Explain why each of these three things causes the magnesium melting
and boiling points to be higher.
b) Explain why metals are good conductors of electricity.
c) Explain why metals are also good conductors of heat.
1. Magnesium has a higher melting and boiling point than sodium. This can
be explained in terms of the electronic structures, the packing, and the
atomic radii of the two elements.
a) Explain why each of these three things causes the magnesium melting
and boiling points to be higher.b) Explain why metals are good conductors of electricity.
c) Explain why metals are also good conductors of heat.
2. Pure metals are usually malleable and ductile.
a) Explain what those two words mean.
b) If a metal is subjected to a small stress, it will return to its original shape
when the stress is removed. However, when it is subjected to a larger
stress, it may change shape permanently. Explain, with the help of simple
diagrams why there is a different result depending on the size of the
stress.
c) When a piece of metal is worked by a blacksmith, it is heated to a high
temperature in a furnace to make it easier to shape. After working it with
a hammer, it needs to be re-heated because it becomes too difficult to
work. Explain what is going on in terms of the structure of the metal.
d) Why is brass harder than either of its component metals, copper andzinc?
SKILLS LAB 12
The figure below shows the electric conductivity of distilled water (A), solid tablesalt (B) and a solution of a table salt (C) respectively.
a) Use the diagrams A, B and C to explain the observations from the set up.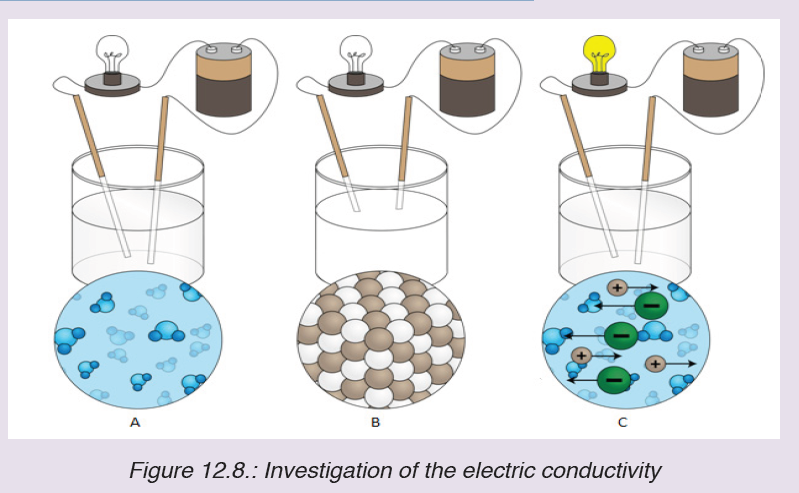
i. No light is given out by bulb in A
ii. No light is given out by bulb in B
iii. Light is given out in C
b) Suppose that you have a 30 cm bar made of table salt. Suggest the
change, if there is any, that can occur and deduce the property related,
when this salt bar is:
i. Dropped from a table of 1 m high to the floor
ii. Immersed in water found in a water bath.c) Dry heated to 100 oC
End unit assessment 12
1. State whether the following statement is True or False. Justify your
answer. “Sodium Chloride has a higher melting point than Magnesium
Oxide”.
2. Why are ionic compounds brittle?
3. Why do ionic compounds have high melting points?
4. What happens when an electric current is passed through a solution of
an ionic compound?
5. This question is about metallic bonding.
a) Describe the bonding that is present in metals.
b) Explain how the bonding and structure lead to the typical metallic
properties of electrical conductivity and malleability.
c) Suggest a reason why aluminium is a better conductor of electricity than
magnesium.
6. Silver and sodium chloride melt at similar temperatures. Give two physical
properties of silver which are different from those of sodium chloride and,
in each case, give one reason why the property of silver is different from
that of sodium chloride.
7. This question is about calcium oxide (CaO).
a) Describe the nature and strength of the bonding in solid calcium oxide.
b) Use the kinetic theory to describe the changes that take place as calcium
oxide is heated from 25°C to a temperature above its melting point.c) State two properties of calcium oxide that depend on its bonding.
